��Ŀ����
����Ŀ�������������ͿƼ��������ش����á�Ϊ�Ƚ�Fe3+��Cu2+�Էֽ�Ĵ�Ч����ij��ѧ�о�С���ͬѧ�ֱ��������ͼ�ס�����ʾ��ʵ�顣��ش�������⣺

(1)���Է�������ͼ��ͨ���۲�______���ԱȽϵó����ۡ�
ͬѧX�۲������֧�Թܲ������ݵĿ������ɴ˵ó�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч���������______(���������������)��������_____________________��
(2)������������ͼ����ʾ��ʵ��ʱ��������40mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԡ�ʵ������Ҫ������������______��
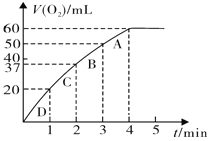
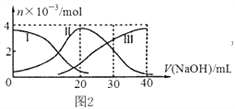
(3)����0.10mol MnO2��ĩ��50mL H2O2��Һ�У��ڱ�״���·ų�����������ʱ��Ĺ�ϵ����ͼ��ʾ��
��ʵ��ʱ�ų�������������______mL��
�ڷų�1/3��������ʱ��Ϊ______ min��
�ۼ���H2O2�ij�ʼ���ʵ���Ũ��______�� (�뱣����λ��Ч����)
��A��B��C��D���㷴Ӧ���ʿ�����˳��Ϊ______��
���𰸡� �������ݵĿ��� ������ ���������ͬ ����40mL���������ʱ�� 60mL 1min 0.11 mol��L-1 D>C>B>A
����������1���÷�Ӧ�в������壬���Կɸ����������ݵĿ����жϣ��Ȼ���������ͭ���������Ӷ���ͬ�����ж��������������û�������������������2���÷�Ӧ��ͨ����������ķ�Ӧ���ʷ����жϵģ����Ը���v=![]() ֪����Ҫ�����������Dz���40ml��������Ҫ��ʱ������3��������ͼ��֪����4����ʱ������ʱ������ƣ�����������䣬����ʵ��ʱ�ų�������������60 mL��������ͼ���֪�ų�1/3���弴����20mL��������ʱ��Ϊ2min������˫��ˮ�����ʵ���Ϊx����
֪����Ҫ�����������Dz���40ml��������Ҫ��ʱ������3��������ͼ��֪����4����ʱ������ʱ������ƣ�����������䣬����ʵ��ʱ�ų�������������60 mL��������ͼ���֪�ų�1/3���弴����20mL��������ʱ��Ϊ2min������˫��ˮ�����ʵ���Ϊx����
2H2O2![]() 2H2O+O2��
2H2O+O2��
2mol 22.4L
x 0.06L
x=![]() =0.0054mol��
=0.0054mol��
���������ʵ���Ũ��=![]() =0.11 mol��L-1��
=0.11 mol��L-1��
����Ӧ�����뷴Ӧ��Ũ�ȳ����ȣ����ŷ�Ӧ�Ľ��У���Ӧ��Ũ�����ͣ���Ӧ������С����˸���ͼ����v=![]() ֪A��B��C��D���㷴Ӧ���ʿ�����˳��ΪD��C��B��A��
֪A��B��C��D���㷴Ӧ���ʿ�����˳��ΪD��C��B��A��