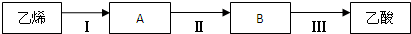��Ŀ����
����йؼصĻ���������⣺
��1������KF��BF3���ÿ�����KBF4��BF4-������ṹΪ ��KBF4�����д��ڵĻ�ѧ���� ��
A�����Ӽ� B�����Լ� C���Ǽ��Լ� D����λ�� E���Ҽ� F���м�
BF3���������� �����ţ���
A��ˮ B���� C�����Ȼ�̼ D��CS2
��2���о����ʴ��Ա��������������Ӻ�δ�ɶԵ���Խ�࣬�����Խ�ż�¼����Խ�ã�������������K2O2��V2O5��MnO2��Fe3O4�У����ʺ���¼�����ŷ�ԭ�ϵ��� ���ѧʽ����
��3���������ɷ���MnO2�����̿���KMnO4��MnO2
K2MnO4
KMnO4��
�ڢ�Ӧ��3MnO42-��aq��+2H2O��l��?2MnO4-��aq��+MnO2��s��+4OH-��aq��������ƽ�ⳣ������ʽK= ��
ͨ��KMnO4�Ƚ��ȶ����ֽ⣬���뱣������ɫƿ�У���Ϊ�չ���ʹ�ֽ⣺
4MnO4-+2H2O
4MnO2��+4OH-+3O2��
һ���ֽ⣬֮�����ʼӿ�ֽ����ԣ����ܵ�ԭ���� ��
��1������KF��BF3���ÿ�����KBF4��BF4-������ṹΪ
A�����Ӽ� B�����Լ� C���Ǽ��Լ� D����λ�� E���Ҽ� F���м�
BF3����������
A��ˮ B���� C�����Ȼ�̼ D��CS2
��2���о����ʴ��Ա��������������Ӻ�δ�ɶԵ���Խ�࣬�����Խ�ż�¼����Խ�ã�������������K2O2��V2O5��MnO2��Fe3O4�У����ʺ���¼�����ŷ�ԭ�ϵ���
��3���������ɷ���MnO2�����̿���KMnO4��MnO2
| �� |
| �� |
�ڢ�Ӧ��3MnO42-��aq��+2H2O��l��?2MnO4-��aq��+MnO2��s��+4OH-��aq��������ƽ�ⳣ������ʽK=
ͨ��KMnO4�Ƚ��ȶ����ֽ⣬���뱣������ɫƿ�У���Ϊ�չ���ʹ�ֽ⣺
4MnO4-+2H2O
| ||
һ���ֽ⣬֮�����ʼӿ�ֽ����ԣ����ܵ�ԭ����
���㣺�жϼ��ӻ����ӵĹ���,��ѧ��,��ѧƽ�ⳣ���ĺ���,��ѧ��Ӧ���ʵ�Ӱ������
ר�⣺��ѧ���뾧��ṹ,��ѧ��Ӧ����ר��
��������1���۲���ӶԻ��������ж���ҵ��ӶԺµ��Ӷ���Ŀ��KF��BF3ͨ����λ����ϣ��ٸ���������������жϣ�������������ԭ��������
��2�����ݷ����ӡ������Ӻ����������������ӵ�δ�ɶԵ����жϣ�
��3������ƽ�ⳣ������ʽ�ĺ�������֪����ʽ��д����Ӧ�������ж������̣����������dz����Ĵ�����
��2�����ݷ����ӡ������Ӻ����������������ӵ�δ�ɶԵ����жϣ�
��3������ƽ�ⳣ������ʽ�ĺ�������֪����ʽ��д����Ӧ�������ж������̣����������dz����Ĵ�����
���
�⣺��1��BF3����NaF���ÿ�����NaBF4��BF4-��Bԭ�ӵļ۲���Ӷ�=4+
��3+1-1��4��=4���������в����µ��Ӷԣ�Ϊ��������ṹ��KF��BF3ͨ����λ����ϣ�KF�д������Ӽ���BF3��B��F����ڼ��Թ��ۼ����ǵ������ڦҼ�����������û��˫��������������û�Цм���������������ԭ�����Ǽ��Է��������ڷǼ����ܼ���BF3�������ڷǼ��Է��ӣ��������Ȼ�̼��CS2���ڷǼ����ܼ���
�ʴ�Ϊ���������壻ABDE��BCD��
��2��K2O2�м�������δ�ɶԵ��ӣ�V2O5�з����ӵĺ�������Ų�ʽΪ��1s22s22p63s23p6����δ�ɶԵ��ӣ�MnO2�������ӵĺ�������Ų�ʽΪ��1s22s22p63s23p63d4����4��δ�ɶԵ��ӣ���Fe3O4����14��δ�ɶԵ��ӣ���Fe3O4�е�δ�ɶԵ�������࣬�ʴ�Ϊ��Fe3O4��
��3����֪��Ӧ��3MnO42-��aq��+2H2O��l��?2MnO4-��aq��+MnO2��s��+4OH-��aq������ƽ�ⳣ������ʽK=
��4MnO4-+2H2O
4MnO2��+4OH-+3O2��
����Ӧ�������ж������̣����������dz����Ĵ�������Ӧ�������ʣ�MnO2���Էֽ��д����ã�ʹKMnO4�ķֽ����ʼӿ죻
�ʴ�Ϊ��
����Ӧ�������ʣ�MnO2���Էֽ��д����ã�
| 1 |
| 2 |
�ʴ�Ϊ���������壻ABDE��BCD��
��2��K2O2�м�������δ�ɶԵ��ӣ�V2O5�з����ӵĺ�������Ų�ʽΪ��1s22s22p63s23p6����δ�ɶԵ��ӣ�MnO2�������ӵĺ�������Ų�ʽΪ��1s22s22p63s23p63d4����4��δ�ɶԵ��ӣ���Fe3O4����14��δ�ɶԵ��ӣ���Fe3O4�е�δ�ɶԵ�������࣬�ʴ�Ϊ��Fe3O4��
��3����֪��Ӧ��3MnO42-��aq��+2H2O��l��?2MnO4-��aq��+MnO2��s��+4OH-��aq������ƽ�ⳣ������ʽK=
| c2(MnO4-)?c4(OH-) |
| c3(MnO42-) |
| ||
����Ӧ�������ж������̣����������dz����Ĵ�������Ӧ�������ʣ�MnO2���Էֽ��д����ã�ʹKMnO4�ķֽ����ʼӿ죻
�ʴ�Ϊ��
| c2(MnO4-)?c4(OH-) |
| c3(MnO42-) |
���������⿼�������Ŀռ乹�͵��жϡ���ѧ������������ԭ���������Ų�ʽ��ƽ�ⳣ������ʽ�ȣ���Ŀ�漰��֪ʶ��϶࣬��Ŀ�Ѷ��еȣ������ڿ���ѧ����֪ʶ���ۺ�Ӧ��������

��ϰ��ϵ�д�
�����Ŀ
��һ���¶��£����淴ӦA������+3B������?2C�������ﵽƽ��ı�־�ǣ�������
| A��C���ɵ�������C�ֽ��������� |
| B��A��B��C��Ũ����� |
| C��A��B��C�ķ�������Ϊ1��3��2 |
| D����λʱ������n mol A��ͬʱ����3n mol B |
���з�Ӧ�У������仯��������ͼͼʾ���ǣ�������

| A���������кͷ�Ӧ |
| B������ʯ��ʯ����ʯ�� |
| C��Ba��OH��2?8H2O��NH4Cl���巴Ӧ |
| D����̿��ˮ������Ӧ |
���б�ʾ��Ӧ��ѧ��Ӧ�����ӷ���ʽ��ȷ���ǣ�������
| A��̼������ڴ��CaCO3+2H+�TCa2++CO2��+H2O | ||||
B����������Һ��ͨ��������CO2��2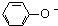 +CO2+H2O�� +CO2+H2O�� +CO32- +CO32- | ||||
C����ͭ���缫���CuSO4��Һ��2Cu2++2H2O
| ||||
D����������Һ������ȩ�е�ȩ����CH3CHO+2Ag��NH3��2++2OH-
|
��������˳����ȷ���ǣ�������
| A�����ȶ���ǿ����HCl��HBr��HI |
| B�����뾶��Cl-��K+��Na+��Fe��OH��3���� |
| C�������۵㣺SiO2��NaCl��CF4��CCl4 |
| D���⻯��ķе㣺H2Se��H2S��H2O |