��Ŀ����
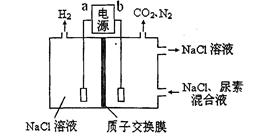
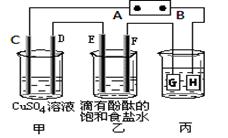
������о���Ա������һ�ִ�����Ũ����ȩ��ˮ���·����� ��Ĥ��ⷨ����ȩ�ֱ�����������������Ӧ��ת��Ϊ�Ҵ������ᡣʵ������һ��Ũ�ȵ���ȩ��Na2SO4��ҺΪ�������Һ��ģ����ȩ��ˮ�Ĵ������̣���װ����ͼ��ʾ������˵����ȷ����

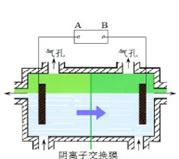
| A������CH4������ȼ�ϵ��Ϊֱ����Դ��ȼ�ϵ�ص�b��Ӧͨ����� |
| B���������У�������Na2SO4�����ʵ������� |
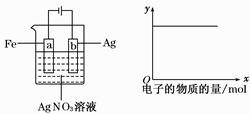
| C��������ӦCH3CHO -2e- + 2H+="=" CH3COOH + H2O |
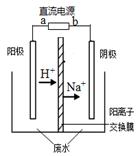
| D���������У��������ֱ�����������Ҵ��⣬����������ɫ���壬��������������O2 |
D
���������A.����CH4������ȼ�ϵ��Ϊֱ����Դ������ͬ�ֵ����ų⣬���ֵ���������ԭ������H+��Na+��������������ȥ�ƶ���˵��aΪ������Ӧͨ�������b�Ǹ�����ͨ��������塣����B. �������У�Na2SO4��������������������缫����������Ӧ���������ʵ������䡣����C. ������ӦӦ��ΪCH3CHO -2e- + H2O ="=" CH3COOH +2H+������D. �������У��������ֱ�����������Ҵ��⣬����������ɫ���壬��������������O2����������H2����ȷ��

��ϰ��ϵ�д�
 Ӧ������ҵ��ϵ�д�
Ӧ������ҵ��ϵ�д�
�����Ŀ

 R2Cu(�л���)��2H��(ˮ��)
R2Cu(�л���)��2H��(ˮ��)