��Ŀ����
20�����мס��ҡ�������ͬѧ�ֱ����Fe��OH��3������Ʊ�ʵ�飮��ͬѧ��1mol•L-1�Ȼ�����Һ�м���������NaOH��Һ��
��ͬѧֱ�Ӽ��ȱ���FeCl3��Һ��
��ͬѧ��25mL��ˮ����μ���1mL��2mL FeCl3������Һ�������������Һ�ʺ��ɫ��ֹͣ���ȣ��Իش��������⣺
��1�����в�����ȷ��ͬѧ�DZ���
��2����֤��Fe��OH��3���强�Ƴɣ����������ü�������䣬����һ��������ͨ·�����н������ɣ���ķ�������
��3����ͬѧ�������Ƶõ�Fe��OH��3�����������ʵ�飺
�ٽ���װ��U�ι��ڣ���ʯī���缫����ֱͨ���磬ͨ��һ��ʱ�����������������ɫ��������Fe��OH��3�����еĽ��������磮
�������м���ϡ���ᣬ�����������������ɺ��ɫ�ij��������ܽ�Ϊ��ɫ��Һ��
��4����ͬѧ�뽫Fe��OH��3�����ᴿ�������Բ��õķ�����������
��5��д����ͬѧ����ʵ������ӷ���ʽFe3++3OH-�TFe��OH��3����
���� ��1���Ʊ�������������ķ�������25ml��ˮ����μ���1��2mL FeCl3������Һ�������������Һ�ʺ��ɫ��ֹͣ���ȣ�
��2�������ЧӦ�ǽ�����������ʣ�
��3���������Ӵ��е�ɣ����е�Ӿ�����ʣ��ܷ����۳���
��4����Һ������Ĥ��������������Ĥ��
��5����Ӧ���������������Ȼ��ƣ�
��� �⣺��1����ͬѧ��1mol•L-�Ȼ�����Һ�м���������NaOH��Һ���ᷢ�����ֽⷴӦ���ɺ��ɫ��������ͬѧֱ�Ӽ��ȱ���FeCl3��Һ����������ˮ�����ɺ��ɫ�������Ʊ�������������ķ�������25ml��ˮ����μ���1��2mL FeCl3������Һ�������������Һ�ʺ��ɫ��ֹͣ���ȣ��ʴ�Ϊ������
��2�������ЧӦ�ǽ�����������ʣ������������壬�ʴ�Ϊ���ü�������䣬����һ��������ͨ·�����н������ɣ�
��3����Fe��OH��3���������磬ͨ��ʱ������ɵ������������ƶ�������������ɫ����ʴ�Ϊ������
�ڽ���������ʷ����۳����������������ᷴӦ�������������ʴ�Ϊ�������ɺ��ɫ�ij��������ܽ�Ϊ��ɫ��Һ��
��4����Һ������Ĥ��������������Ĥ�������������ᴿ���壬�ʴ�Ϊ��������
��5����Ӧ���������������Ȼ��ƣ����ӷ���ʽ��Fe3++3OH-�TFe��OH��3�����ʴ�Ϊ��Fe3++3OH-�TFe��OH��3����
���� ���⿼�齺����Ʊ������ʣ���Ŀ�ѶȲ����״���Ϊ������Ʊ���ע���Ʊ������������ص���ս���ľ۳������ʣ�

| A�� | H3PO4��Ħ������Ϊ98 | |
| B�� | ����٤��������Ϊ0.012kg12C������̼ԭ���� | |
| C�� | �κ������Ħ�������ԼΪ22.4L��mol-1 | |
| D�� | һ��������ѹ��4��ʱ��9mLˮ������������ԼΪ3.01��1024�� |
| A�� | �����Ƽ�Ĺ��չ�����Ӧ���������ܽ�ȵIJ��� | |
| B�� | �������������Ԫ�أ����ҹ涨ʳ�α���ӵ⣮�ʳ���ʱӦ�ڲ˿����ʱ���αȽϺ� | |
| C�� | ���ʹ賣�������ά�IJ��ϣ������İ뵼��������һ����ϵ | |
| D�� | ��ѹ�ƵƷ����Ļƹ����Զ��������ǿ��������·�� |
| A�� | ���ǻ��Ϸ�Ӧ | |
| B�� | �ڱ��ɫ����Ϊ������̼���� | |
| C�� | ����̼�������տ����е�ˮ�����γ�����Һ | |
| D�� | ��ֻ���������仯 |
| A�� | ��״���£�11.2L�Ҵ��к��е�̼�����Ϊ2.5NA | |
| B�� | ���³�ѹ�£�28gC2H4��CO�Ļ�������к���̼ԭ�ӵ���ĿΪ1.5NA | |
| C�� | �����£�20LpH=12��Na2CO3��Һ�к��е�OH-������Ϊ0.2NA | |
| D�� | ��״���£�2.24LCl2��������NaOH��Һ��Ӧ��ת�Ƶĵ�����ĿΪ0.2NA |
| A�� | ����֬�� | B�� | ��������ά�� | C�� | 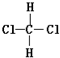 �� ��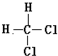 | D�� | ��������춡�� |
| A�� | �����ʵ�����N2��O2�е�������������ΪNA | |
| B�� | ��״���£�5.6L CCl4���еķ�����Ϊ0.25NA | |
| C�� | ���³�ѹ�£�22gCO2���е�ԭ����Ϊ1.5NA | |
| D�� | 1mol/L NaOH��Һ�к��е�Na+����ĿΪNA |
 ��0.1mol MnO2��ĩ���뵽50mL����������Һ��H2O2����=1.1g/mL���У��ڱ�״���·ų�����������ʱ��Ĺ�ϵ��ͼ��ʾ��
��0.1mol MnO2��ĩ���뵽50mL����������Һ��H2O2����=1.1g/mL���У��ڱ�״���·ų�����������ʱ��Ĺ�ϵ��ͼ��ʾ��