��Ŀ����
����������д�����з�Ӧ���Ȼ�ѧ����ʽ��
��1����NA��ʾ�����ӵ���������C2H2(��̬)��ȫȼ������CO2��Һ̬ˮ�ķ�Ӧ�У�ÿ��4NA������ת��ʱ���ų�450 kJ�����������Ȼ�ѧ����ʽΪ______________________��
��2����֪��1 mol H��H����1 mol N��H����1 mol N��N���ֱ���Ҫ��������436 kJ��395 kJ��940 kJ����N2��H2��Ӧ����NH3���Ȼ�ѧ����ʽΪ____________________________��
��3���ѣ�Ti������Ϊ��������֮��ĵ�����������֪�ɽ��ʯ��TiO2����ȡ����Ti���漰�IJ���Ϊ��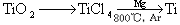
��֪����C(s)+O2(g) 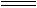 CO2(g); ��H=��395.5 kJ��mol-1
CO2(g); ��H=��395.5 kJ��mol-1
��2CO(g)+O2(g) 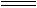 2CO2(g); ��H=��560 kJ��mol-1
2CO2(g); ��H=��560 kJ��mol-1
��TiO2(s)+2Cl2(g)+2C(s) 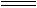 TiCl4(s)+2CO(g)�Ħ�H=�D80kJ/mol
TiCl4(s)+2CO(g)�Ħ�H=�D80kJ/mol
��TiO2(s)��Cl2(g)��Ӧ���Ȼ�ѧ����ʽΪ ��
��13�֣�
(1)C2H2(g)+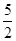 O2(g)��2CO2(g)+ H2O(l) ��H����1125 kJ/mol ��4�֣�
O2(g)��2CO2(g)+ H2O(l) ��H����1125 kJ/mol ��4�֣�
(2)N2(g)+ 3H2(g)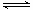 2NH3(g) ��H����122 kJ/mol ��4�֣�
2NH3(g) ��H����122 kJ/mol ��4�֣�
(3)TiO2(s)+ 2Cl2(g)��TiCl4(s)+ O2(g) ��H����151 kJ/mol ��5�֣�
���������������1����Ȳ������̼Ԫ�صĻ��ϼ��ǣ�1�ۣ���Ӧ���Ϊ��4�ۣ�ʧȥ5�����ӣ���1mol��Ȳʧȥ10mol���ӣ���ÿ��4NA������ת��ʱ��������Ȳ�����ʵ�����0��4mol������ÿ����1mol��Ȳ�ų���������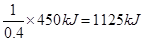 ����˸÷�Ӧ���Ȼ�ѧ����ʽ��C2H2(g)+
����˸÷�Ӧ���Ȼ�ѧ����ʽ��C2H2(g)+ 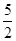 O2(g)��2CO2(g)+ H2O(l) ��H����1125 kJ/mol��
O2(g)��2CO2(g)+ H2O(l) ��H����1125 kJ/mol��
��2����Ӧ�Ⱦ��Ƕϼ����յ����������γɻ�ѧ�����ų��������IJ�ֵ������ݼ��ܿ�֪��ÿ����2mol�����ķ�Ӧ�ȡ�H��436kJ/mol��3��940kJ/mol��2��3��395kJ/mol����122 kJ/mol������Ӧ���Ȼ�ѧ����ʽ��N2(g)+ 3H2(g)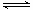 2NH3(g) ��H����122 kJ/mol��
2NH3(g) ��H����122 kJ/mol��
��3�����ݸ�˹���ɿ�֪���ۣ��ڣ��١�2�����õ���ӦTiO2(s)+ 2Cl2(g)��TiCl4(s)+ O2(g) �����Ը÷�Ӧ�ķ�Ӧ�Ȧ�H���D80kJ/mol��560 kJ/mol��395��5 kJ/mol��2����151 kJ/mol��
���㣺�����Ȼ�ѧ����ʽ����д�Լ���Ӧ�ȵ��йؼ��㡣

��֪����1 mol H2�����л�ѧ������ʱ��Ҫ����436 kJ����������1 mol I2�����л�ѧ������ʱ��Ҫ����151 kJ������������Hԭ�Ӻ�Iԭ���γ�1 mol HI��̬����ʱ�ͷ�299 kJ�������������Ȼ�ѧ����ʽ��ȷ����
| A��2HI(g) =H2(g)��I2(g)?H��+11 kJ��mol |
| B��H2(g)��I2(g) =2HI(g)?H����22 kJ��mol |
| C��H2(g)��I2(g) =2HI(g)?H��+288 kJ��mol |
| D��H2(g)�� I2(g) =2HI(g)?H����144 kJ��mol |
��֪��2H2(g)��O2(g)=2H2O(g) ��H����483.6 kJ��mol��1
H2(g)��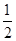 O2(g)=H2O(l) ��H����285.8 kJ��mol��1
O2(g)=H2O(l) ��H����285.8 kJ��mol��1
�ɴ˿�֪���ڵ���������36 gҺ̬ˮ�����յ������� ��
| A��483.6 kJ | B��88 kJ | C��285.8 kJ | D��44 kJ |
���������仯ʾ��ͼ������ѡ����ȷ����
| A��S��s�����+O2��g��=SO2��g����H ="+297.16" kJ?mol-1 |
| B��S��s�����+O2��g��=SO2��g����H ="-296.83" kJ?mol-1 |
| C��S��s��������+O2��g��=SO2��g����H ="-296.83" kJ?mol-1 |
| D����б�����������ȶ� |
���з�Ӧ�оɻ�ѧ���������յ������������»�ѧ���γɷų�����������
| A�����ˮ��ȡH2��O2 | B������ȼ�� |
| C����������������ĩ��Ӧ | D����֬����������ȫ��������CO2��H2O |
�γɽ�Լ��Դ�ͱ�����̬�����IJ�ҵ�ṹ����������Ȼ��г��չ����Ҫ��֤������Ϊ������Ϊ���������һ��֤���ǣ� ��
| A���о���ú�������¼�������߲��������㹤ҵ�����Ŀ��ٷ�չ |
| B������̫���ܡ�ˮ�ܡ����ܵ�����Դ������ʹ��ú��ʯ�͵Ȼ�ʯȼ�� |
| C����ũ���ƹ�ʹ������ |
| D��������Դ���ġ�������Դ���ظ�ʹ�ú���Դ��ѭ������ |
���з�Ӧ�е������仯��ϵ������ͼ��ʾ����
| A���������ռӦ |
| B����Ȼ��ȼ�� |
| C������������ˮ��Ӧ |
| D������ʯ��ʯ |
��֪2H2(g)+O2(g) ��2H2O(l����H����571.6kJ��mol��1��2H2(g)+O2(g)��2H2O(g����H����483.6kJ��mol��1������˵����ȷ����
| A��1molH2O(l)���1mo1H2O(g)������88 kJ���� |
| B��1 molH2O(g)�ֽ��H2(g)��O2 (g)������241.8kJ���� |
| C��2 molH2(g)��1molO2(g)����������2molH2O(l)������ |
| D������Ѱ���������ʹH2O�ֽ⣬ͬʱ�ų����� |
�����й�˵����ȷ����
| A��NH4Cl(s)��NH3(g)+HCl(g)�����²����Է����У�˵���÷�Ӧ�Ħ�H��0 |
| B����п����Ʒ�Ʋ����������Ʒ������ǰ���������⣬�����������෴ |
C������N2(g)+3H2(g) 2NH3(g)����������������ʱ��ѹ���������ʹѹǿ��������Ӧ���淴Ӧ�����Լ�H2��ƽ��ת���ʾ����� 2NH3(g)����������������ʱ��ѹ���������ʹѹǿ��������Ӧ���淴Ӧ�����Լ�H2��ƽ��ת���ʾ����� |
| D��100��ʱˮ�����ӻ�����KwΪ5.5��10��13��˵��ˮ�ĵ����Ƿ��ȷ�Ӧ |