��Ŀ����
����С������9�֣� ��ͼ��ʾ�����ס�������װ�в�ͬ���ʵ���Ͳ�õ�������������������Ͳ�ڵ�����ѹ������Ͳ�ڣ������±����еIJ�ͬʵ�飨������ͬ��ͬѹ�²ⶨ�����Իش��������⣺
��1��ʵ��1����Ͳ�ڵ������ǣ���___________���ɣ���Ͳ����________�ƶ��������⡢���ڡ���������Ӧ�����Ͳ���������IJ������壬��ȷ�Ĵ��������ǽ���ͨ��___________��Һ�С�
��2��ʵ��2�У����е�3mL������NO2��N2O4�Ļ�����壬��ô�������ʣ�����ɫ������_______��д��NO2��H2O��Ӧ�Ļ�ѧ����ʽΪ____________________��
��3��ʵ��3�У���֪��3Cl2��2NH3===N2��6HCl������Ͳ���������ƶ�����Ͳ���а��̲����⣬�������ɫ��Ϊ____________�������Ͳ��ʣ����������ԼΪ________mL��
��1����ɫ���壻���ڣ�NaOH����ÿ��1�֣�
��2��NO ��1�֣��� 3NO2+H2O=2HNO3��NO��2�֣���
��3����ɫ��1�֣��� 5��2�֣�
����:��1��SO2��H2S����Ԫ�صĻ��ϼ۷ֱ��ǣ�4�ۺͣ�2�ۣ����Զ��߿��Է���������ԭ��Ӧ���ɵ������ˮ������ʽΪSO2��2H2S=3S��2H2O�����Է�Ӧ��ѹǿ���ͣ���Ͳ���������ƶ���SO2��H2S�����ڴ�����Ⱦ���Ҫβ�����������ü�Һ�����ա�
��2��NO2����ˮ�ķ���ʽΪ3NO2+H2O=2HNO3��NO���������յ���������ɫ��NO��
��3���������Խ������������ɵ���������������ԭ�����Ȼ��⡣���ݷ���ʽ���йط�Ӧ��������֪��15ml������������10ml����������5ml������30ml�Ȼ��⡣������ʣ��30ml�������İ������Ȼ���ǡ�÷�Ӧ�����Ȼ�泥��Ӷ����ְ�������������յ�������5ml������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
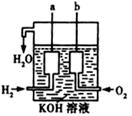
Сѧ��10����Ӧ����ϵ�д�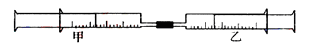

 2C(g)+xD(g)����5min�ﵽƽ�⣬��ʱ����2 mol C�����D��ƽ����Ӧ����Ϊ0.15 mol/(L��min)����ƽ��ʱA�����ʵ���Ũ����____________��B��ת������__________��x��ֵ��___________��
2C(g)+xD(g)����5min�ﵽƽ�⣬��ʱ����2 mol C�����D��ƽ����Ӧ����Ϊ0.15 mol/(L��min)����ƽ��ʱA�����ʵ���Ũ����____________��B��ת������__________��x��ֵ��___________��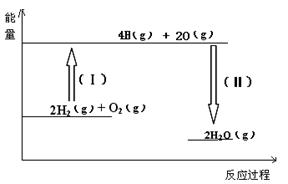
 2C(g)+xD(g)����5min�ﵽƽ�⣬��ʱ����2 mol C�����D��ƽ����Ӧ����Ϊ0.15 mol/(L��min)����ƽ��ʱA�����ʵ���Ũ����____________��B��ת������__________��x��ֵ��___________��
2C(g)+xD(g)����5min�ﵽƽ�⣬��ʱ����2 mol C�����D��ƽ����Ӧ����Ϊ0.15 mol/(L��min)����ƽ��ʱA�����ʵ���Ũ����____________��B��ת������__________��x��ֵ��___________��


