��Ŀ����
����Ŀ���±���Ԫ�����ڱ���һ����, ��Ա��еĢ١�����Ԫ��,��д���пհ�:
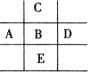
�� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0�� |
2 | �� | �� | �� | |||||
3 | �� | �� | �� | �� | �� | |||
4 | �� |
(1)����ЩԪ���У���ѧ������������ǣ�_______(������Ԫ�ط��Ż�ѧʽ,��ͬ)��
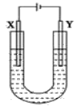
(2) ������������ˮ�����У�������ǿ�Ļ��������ʽ��_________�����������м�����ǿ�Ļ�����Ļ�ѧʽ��____________________��
(3)����������������������Ԫ����____________��д��������������Ӧˮ�������������Ʒ�Ӧ����ѧ����ʽ_______________________________________��
(4)�õ���ʽ��ʾԪ�آ�����γɻ�������γɹ��̣�________________________���û���������__________________(�� ������������������)�����
(5)��ʾ��������̬�γɵĻ���������ʽ___________________���û����������___________(�������������Ǽ�����)���γɡ�
���𰸡�Ar HClO4 NaOH Al Al(OH)3 + NaOH = NaAlO2 + 2H2O ![]() ����
���� ![]() ����
����
��������
��Ԫ�������ڱ���λ�ã���֪��ΪC����ΪN����ΪO����ΪNa����ΪAl����ΪS����ΪCl����ΪAr����ΪK��ͬ���ڴ�����Ԫ�ؽ��������μ������ǽ�����������ǿ��ͬ����Ԫ���д��ϵ��£�Ԫ�ؽ�����������ǿ���ǽ��������μ������ҷǽ�����Խǿ����Ӧ������������Ӧ��ˮ����������Խǿ��������Խǿ����Ӧ������������Ӧ��ˮ�����м���Խǿ���ٽ��Ԫ�������������ʵĽṹ�����ʽ��з������
������������֪����ΪC����ΪN����ΪO����ΪNa����ΪAl����ΪS����ΪCl����ΪAr����ΪK��
��1��0��Ԫ�ػ�ѧ���ʲ����ã����ȶ���������Ԫ���У���ѧ�������������Ar��
�ʴ�Ϊ��Ar��
��2���ǽ�����Խǿ��������������ˮ����������Խǿ����������ǿ����HClO4��������Խǿ��������������Ӧ��ˮ�������Խǿ������������м�����ǿ�Ļ�����ΪNaOH��
�ʴ�Ϊ��HClO4��NaOH��
��3���������ȿ������ᷴӦ�����κ�ˮ���ֿ�����Ӧ��������ˮ��������������������������Ԫ��ΪAl��������������Ӧˮ����Ϊ�������������������Ʒ�Ӧ����ѧ����ʽΪAl(OH)3 + NaOH = NaAlO2 + 2H2O��
�ʴ�Ϊ��Al��Al(OH)3 + NaOH = NaAlO2 + 2H2O��
��4��Ԫ�آ�����γɻ�����Na2S��Ϊ���ӻ�������õ���ʽ��ʾ���γɹ���Ϊ��![]() ��
��
�ʴ�Ϊ��![]() �����ӣ�
�����ӣ�
��5����������̬�γɵĻ����������̼��Ϊ���ۻ���������ں�̼�����Լ��������ʽΪ��![]() ��
��
�ʴ�Ϊ��![]() �����ԡ�
�����ԡ�

 �Ͻ�ƽ��У����ϵ�д�
�Ͻ�ƽ��У����ϵ�д�