��Ŀ����
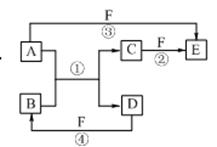
����ת����ϵ�У�X��Y����������;�㷺�����ֽ������ʣ�A��B�������A�ʺ���ɫ��C��D��E����ѧ���������ֻ��������ת����ϵ�ش����⣺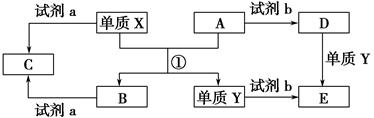
(1)��д����Ӧ�ٵĻ�ѧ����ʽ_____________________________________��
(2)����D��Һ��Y���ӵķ�����__________________________________��
(3)���Լ�a��NaOH��Һ��д������X��NaOH��Һ��Ӧ�����ӷ���ʽ______________________________��
(4)���Լ�b��H2SO4����ҵ����E��H2SO4��NaNO2Ϊԭ����ȡ��Ч��ˮ��Y(OH)SO4����֪��ԭ����ΪNO����÷�Ӧ�Ļ�ѧ����ʽ��________________________________________��
(5)��ҵ�ϵ�����ڵ�B��ȡXʱ�������������������ڱ�״���µ����Ϊ33.6 m3�����������������Ϊ________kg��
��(1)Fe2O3��2Al 2Fe��Al2O3��(2)ȡ����D��Һ���Թ��У��μӼ���KSCN��Һ������Һ���ɫ����֤����Fe3����(3)2Al��2OH����2H2O===2AlO2����3H2��
2Fe��Al2O3��(2)ȡ����D��Һ���Թ��У��μӼ���KSCN��Һ������Һ���ɫ����֤����Fe3����(3)2Al��2OH����2H2O===2AlO2����3H2��
(4)2FeSO4��H2SO4��2NaNO2===2Fe(OH)SO4��Na2SO4��2NO����(5)54
����

 �����Ƹ���ʦ����ϵ�д�
�����Ƹ���ʦ����ϵ�д� ��ͨ����ͬ����ϰ��ϵ�д�
��ͨ����ͬ����ϰ��ϵ�д��ס��ҡ����������������У��ס��ҡ�����������ͬ��ij��Ԫ�أ�����֮���������ת����ϵ��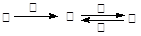 �������й����ʵ��ƶϲ���ȷ����(�� ��)
�������й����ʵ��ƶϲ���ȷ����(�� ��)
| A������Ϊ��̿��������O2 | B������ΪSO2�������ǰ�ˮ |
| C������ΪFe������������ | D������ΪNaOH ��Һ��������AlCl3 |
A~H��Ϊ������Ԫ�أ�A~F��Ԫ�����ڱ��е����λ����ͼ1��ʾ��G����������Ԫ��
| ͼ1 | |||
| A | B | C | |
| D | | E | F |
| ͼ2 | |||
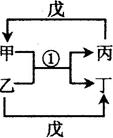 | |||
����ͬһ���ڣ�H�Ƕ�������ԭ�Ӱ뾶��������Ԫ�ء���B��G���ɵ�������ﳣΪ���ʹ�ҵ�ʹ��ҵ��ԭ�ϡ�������ijЩԪ����ɵ����ʼ�~���ת����ϵ��ͼ2��ʾ����ش��������⣺
��1����֪ͼ2�з�Ӧ���Ǹ��ֽⷴӦ����������ˮ����ȥ��
a�������Ǻ���18���ӵ�˫ԭ�ӷ��ӣ���ĵ���ʽΪ��_________��ʵ������ȡ������Ļ�ѧ����ʽΪ��___________________________��
b������ˮ��Һ�ʼ��ԣ�����������������ˮ�������������д����һ����; ��
c������a��b�мı�����Һ��ϣ�����ְ�ɫ��״������ͬʱ���д̼�����ζ�������������÷�Ӧ�����ӷ���ʽ�� ��
��2����֪ͼ2�з�Ӧ�����û���Ӧ�����ǵ��ʣ���������� �� ���ֱ�д��һ����Ӧ�ķ�Ӧ�ٵĻ�ѧ����ʽ �� ��
��.���л������У����зǼ��Թ��ۼ������ӻ������� (����)��
| A��CaC2 | B��N2H4 | C��Na2S2 | D��NH4NO3 |
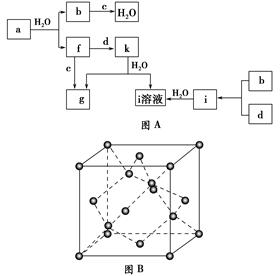
�ش��������⣺
(1)ͼB��Ӧ������������________���侧���е�ԭ����Ϊ________����������Ϊ________��
(2)d��Ԫ�ص�ԭ�Ӻ�������Ų�ʽΪ________��
(3)ͼA���ɶ���Ԫ����ɵ������У��е���ߵ���________��ԭ����________________________________________________________________________�������ʵķ��ӹ���Ϊ________������ԭ�ӵ��ӻ��������Ϊ________��
(4)ͼA�е�˫ԭ�ӷ����У��������ķ�����________��
(5)k�ķ���ʽΪ________������ԭ�ӵ��ӻ��������Ϊ________������________����(����ԡ��Ǽ��ԡ�)��