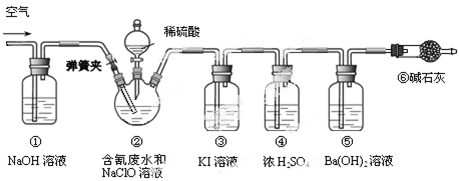
��Ŀ����
(17��)Na2SO3�����ڿ����о������ױ��� ��
(I)Na2SO3�����ڿ����б��ʵ���Ҫԭ���û�ѧ����ʽ��ʾΪ ��
(II)Ϊ̽��Na2SO3��Ʒ�ı����������������¼��裺
����1��Na2SO3��Ʒ��ȫ���ʣ� ����2��Na2SO3��Ʒ��ȫû�б��ʣ�����3�� ��
�����������ʵ����̼�����ͽ��ۣ����̽����
| ʵ����� | ����ͽ��� |
| ����1��ȡ������Ʒ���Թ��У�������������ˮ����ܽ⣬�ٵμ�H2SO4�ữ��KMnO4��Һ�� | ������KMnO4��Һ���Ϻ�ɫ��Ϊ��ɫ �ٽ��ۣ���Ʒ���� ���ӣ�����1�������� ����ɫ�����ӷ���ʽΪ�� �� |
| ����2����ȡ������Ʒ���Թ��У�������������ˮ����ܽ⣬�ٵμ�ϡHCl��ʹ��Һ�����ԣ��ٵμ�����BaCl2��Һ�� | �������� �� ���ۣ�����2������ |
| ���� | ���� |
���Bװ�������ԵIJ���Ϊ ��
��2��ʵ���д���ƿ�в��ٲ����������P�ӵ�����˻�������һ�����Ŀ�������������Ŀ���� ��
��3�����ѳ�����a g Na2SO3��Ʒ�⣬ʵ���л�Ӧ�ⶨ�������� ��(��ͼ����ĸ)װ��ʵ��ǰ��������
(I) 2Na2SO3+ O2 ==2Na2SO4��
(II)����3�� Na2SO3���ֱ��� ����SO32-���ӣ� ��5SO32-+2MnO4-+ 6H+ =5SO42- +2Mn2++3H2O��
��������������������壬��BaCl2�ް�ɫ�������� ��
(III)��1�� �رտ���P�ͷ�Һ©����������C�м�ˮ����û���ܿڣ���B��C���ܿ�������ð����ֹͣ����һ��������γɵ�����ˮ���������������á�
��2�� ��װ���е�SO2ȫ������U�ι������ա� ��3�� D ��
�������������(I)��Na2SO3��S�Ļ��ϼ�Ϊ+4�ۣ����ױ������е���������Ϊ+6�۵�Na2SO4.��Ӧ�ķ���ʽ��2Na2SO3+ O2 ==2Na2SO4��(II)������Ŀ�Ѿ�������֪ʶ��֪������3��Na2SO3���ֱ��ʣ���H2SO4�ữ��KMnO4��Һ��ǿ�����ԣ����뻹ԭ�Ե����ӷ�����Ӧ����ɫ�����ۣ���Ʒ����SO32-���ӣ�֤������1���������ڷ�����ɫ�����ӷ���ʽΪ��5SO32-+2MnO4-+ 6H+ =5SO42- +2Mn2++3H2O����ȡ������Ʒ���Թ��У�������������ˮ����ܽ⣬�ٵμ�ϡHCl��ʹ��Һ�����ԣ��ٵμ�����BaCl2��Һ������������������壬��BaCl2�ް�ɫ�������ɣ���֤������SO32-��SO42-����.����2������(III)��1�����Bװ�������ԵIJ���Ϊ�رտ���P�ͷ�Һ©����������C�м�ˮ����û���ܿڣ���B��C���ܿ�������ð����ֹͣ����һ��������γɵ�����ˮ���������������á���2��ʵ���д���ƿ�в��ٲ����������P�ӵ�����˻�������һ�����Ŀ�������������Ŀ���ǽ�װ���е�SO2ȫ������U�ι������գ��Լ�Сʵ������3�����ѳ�����a g Na2SO3��Ʒ�⣬ʵ���л�Ӧ�ⶨ��������D��U�ι���������ǰ���������ֵ��
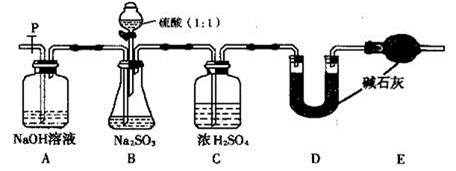
���㣺����װ�õ������Եļ�顢���ӵļ��顢���ӷ���ʽ����д��ʵ�鷽������ơ��о������۵�֪ʶ��

Ũ����������У������ԡ���ǿ�����ԡ�����ˮ�ԡ����ѻӷ��Եȡ��Ҵ����廯�ƺ�Ũ�����Ϲ����Ʊ�������ʱ�����ж������Ӧ�������˹�����Ũ������ʾ��������
| A���٢ڢۢ� | B���ٺ͢� | C��ֻ�Т� | D���ٺ͢� |
һͬѧ������Ԫ�صĻ��ϼ��У�2��0����4����6�������ʴ����м��̬��������Ӧ�ü��������ԣ����л�ԭ�ԡ���ͬѧ��̽����Ļ�ԭ�ԣ������Ǹ�ͬѧ��ʵ��̽�����̣��������е����⡣
(1)��ͬѧӦ��ѡ��________(�����������ԭ����)����ʵ��̽����
(2)��ͬѧ�ú��ȵIJ�������ȼ��ʯ�����ϵ���ۣ���ۿ�ʼȼ�գ���Ӧ�Ļ�ѧ����ʽΪ________________________________________________
(3)��ͬѧ����֤��ȼ�ղ�������ʣ����������ѡ����ʵ��Լ�����˵��ԭ��
| A�����Ƶ���ˮ | B��Ʒ����Һ | C�����з�̪��NaOHϡ��Һ | D��H2S���� |
| ���� | �Լ� | ��ѧ����ʽ |
| Ư���� | | |
| ������ | | |
| ��ԭ�� | | |
| ���������� | | |

 2Ag2O��4NO2����O2�� ����2AgNO3
2Ag2O��4NO2����O2�� ����2AgNO3