��Ŀ����
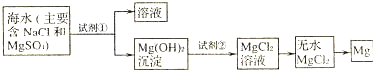
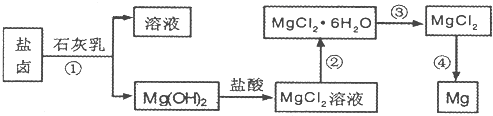
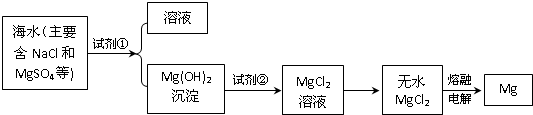
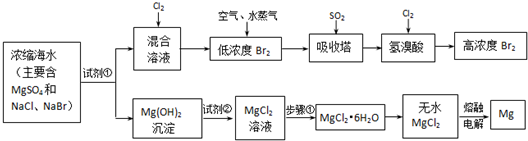
Ŀǰ������60%��þ�ǴӺ�ˮ����ȡ�ģ�ѧ�����������չ�������۵ģ���֪��ˮ��þ����Ҫ������ͼ��ʾ��
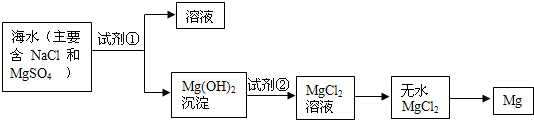
ѧ�����������������������⣺
��һ���ں�ˮ��þ�Ĺ��������ʵ�ֶ�þ���ӵĸ�����������ѧ������Լ��Ĺ۵㣮
ѧ��1�Ĺ۵㣺ֱ������ˮ�м����������
ѧ��2�Ĺ۵㣺���¼���������ˮ���ټ����������
ѧ��3�Ĺ۵㣺����ɹ�κ�Ŀ�±ˮ���ټ����������
������������ѧ������Ĺ۵��Ƿ���ȷ�����ǻ�����������ɣ�
�������ں�ˮ��þ�Ĺ��������ʵ�ֶ�þ���ӵķ��룿
��1��Ϊ��ʹþ���ӳ�������������������Լ�����
��2���Դӽ�Լ��Դ����߽���þ�Ĵ��ȷ���������ұ��þ�ķ����ǣ��û�ѧ����ʽ��ʾ��

ѧ�����������������������⣺
��һ���ں�ˮ��þ�Ĺ��������ʵ�ֶ�þ���ӵĸ�����������ѧ������Լ��Ĺ۵㣮
ѧ��1�Ĺ۵㣺ֱ������ˮ�м����������
ѧ��2�Ĺ۵㣺���¼���������ˮ���ټ����������
ѧ��3�Ĺ۵㣺����ɹ�κ�Ŀ�±ˮ���ټ����������
������������ѧ������Ĺ۵��Ƿ���ȷ�����ǻ�����������ɣ�
| �Ƿ���ȷ | �������� | |
| ѧ��1�Ĺ۵� | �� �� |
��ˮ��þ����Ũ��С��������������������þ���ӵij��� ��ˮ��þ����Ũ��С��������������������þ���ӵij��� |
| ѧ��2�Ĺ۵� | �� �� |
��Դ���Ĵ�ˮ���ۺ����õͣ��ɱ��� ��Դ���Ĵ�ˮ���ۺ����õͣ��ɱ��� |
| ѧ��3�Ĺ۵� | �� �� |
þ���Ӹ���Ũ�ȸߣ��ɱ��� þ���Ӹ���Ũ�ȸߣ��ɱ��� |
��1��Ϊ��ʹþ���ӳ�������������������Լ�����
NaOH
NaOH
���ѧʽ������2���Դӽ�Լ��Դ����߽���þ�Ĵ��ȷ���������ұ��þ�ķ����ǣ��û�ѧ����ʽ��ʾ��
Mg��OH��2+2HCl=MgCl2+2H2O��MgCl2�����ڣ�
Mg+Cl2��
| ||
Mg��OH��2+2HCl=MgCl2+2H2O��MgCl2�����ڣ�
Mg+Cl2��
��
| ||
��������һ�����ԱȽ������۵���ŵ��ȱ�㣬�ۺϷ�����
��������1���ӷ����Ļ�ѧ��Ӧ��������
��2������ȡ����þ�Ĺ��̿��ǣ�
��������1���ӷ����Ļ�ѧ��Ӧ��������
��2������ȡ����þ�Ĺ��̿��ǣ�
����⣺��һ��ѧ��1�������۵���ȣ���ˮ��þ����Ũ��С��ʹ�õij����������ϴ��Ҳ������ռ�������þ�����ѧ��1�Ĺ۵㲻��ȷ��ԭ���ǣ���ˮ��þ����Ũ��С��������������������þ���ӵij�����
ѧ��2�������۵���ȣ�����������ˮ�����ĵ���Դ�࣬�ɱ�̫�ߣ����ѧ��2�Ĺ۵㲻��ȷ��ԭ���ǣ���Դ���Ĵ�ˮ���ۺ����õͣ��ɱ��ߣ�
ѧ��3��ѧ��2��ȣ�����ɹ�κ�Ŀ�±����Լ��Դ���ɱ��ͣ���ѧ��1���þ����Ũ�ȸߣ�������þԪ�صĸ��������ѧ��3�Ĺ۵���ȷ����Ϊ��þ���Ӹ���Ũ�ȸߣ��ɱ��ͣ�
�ʴ�Ϊ��
��������1�������Լ��ٷ�Ӧ����������þ��ʣ����Һ�����ʹ���������ƣ�������������þ�ֲ���ʣ����Һ�����������������ӣ���Ȼ����������������Һ����;������ȡ�Ȼ��ƣ��������Ǿ���Ч�棬��Ҫʹ���������ƣ��ʴ�Ϊ��NaOH��
��2����ȡþʱ��Ҫ��������þת��Ϊ��ˮ�Ȼ�þ��Ȼ��������״̬���Ȼ�þ����Ӧ����ʽ��Mg��OH��2+2HCl=MgCl2+2H2O��MgCl2�����ڣ�
Mg+Cl2�����ʴ�Ϊ��Mg��OH��2+2HCl=MgCl2+2H2O��MgCl2�����ڣ�
Mg+Cl2����
ѧ��2�������۵���ȣ�����������ˮ�����ĵ���Դ�࣬�ɱ�̫�ߣ����ѧ��2�Ĺ۵㲻��ȷ��ԭ���ǣ���Դ���Ĵ�ˮ���ۺ����õͣ��ɱ��ߣ�
ѧ��3��ѧ��2��ȣ�����ɹ�κ�Ŀ�±����Լ��Դ���ɱ��ͣ���ѧ��1���þ����Ũ�ȸߣ�������þԪ�صĸ��������ѧ��3�Ĺ۵���ȷ����Ϊ��þ���Ӹ���Ũ�ȸߣ��ɱ��ͣ�
�ʴ�Ϊ��
| �Ƿ���ȷ | �������� | |
| ѧ��1�Ĺ۵� | �� | ��ˮ��þ����Ũ��С��������������������þ���ӵij����� |
| ѧ��2�Ĺ۵� | �� | ��Դ���Ĵ�ˮ���ۺ����õͣ��ɱ��ߣ� |
| ѧ��3�Ĺ۵� | �� | þ���Ӹ���Ũ�ȸߣ��ɱ��ͣ� |
��2����ȡþʱ��Ҫ��������þת��Ϊ��ˮ�Ȼ�þ��Ȼ��������״̬���Ȼ�þ����Ӧ����ʽ��Mg��OH��2+2HCl=MgCl2+2H2O��MgCl2�����ڣ�
| ||
| ||
���������⿼��þ���Ʊ�����ȷ��ˮ��þ���Ʊ������̼��Լ���ѡ�����������ע��MgO���۵����ѧ����������Եĵط����Ʊ�þ���õ�ⷨ��

��ϰ��ϵ�д�
�����Ŀ
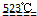 MgO+2 HCl��+5H2O��
MgO+2 HCl��+5H2O��