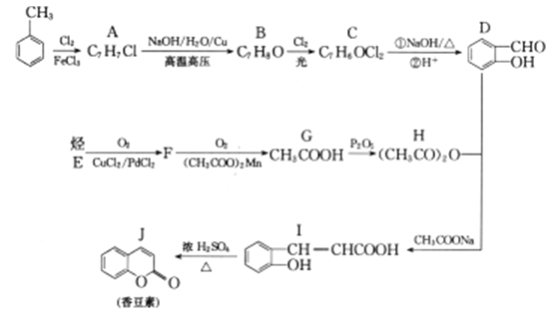
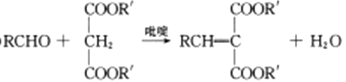
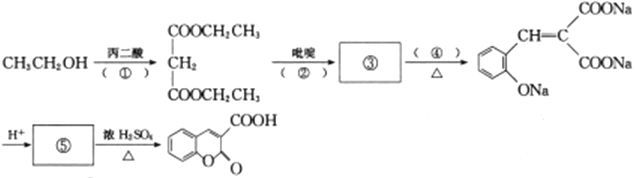
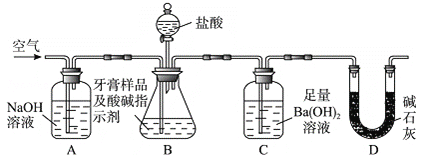
��Ŀ����
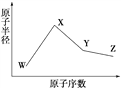
����Ŀ��W��X��Y��Z�����ֳ����Ķ�����Ԫ�أ���ԭ�Ӱ뾶��ԭ�������仯����ͼ��ʾ����֪W��һ�ֺ��ص�������Ϊ18��������Ϊ10��X��Neԭ�ӵĺ�����������1��Y�ĵ�����һ�ֳ����İ뵼����ϣ�Z�ĵ縺����ͬ��������Ԫ�������
(1)Xλ��Ԫ�����ڱ��е�________���ڵ�________�壻W�Ļ�̬ԭ�Ӻ�����_____��δ�ɶԵ��ӡ�
(2)Z����̬�⻯����廯����ȣ����ȶ�����______(д��ѧʽ)��
(3)Y��Z�γɵĻ����������ˮ��Ӧ������һ�������һ��ǿ�ᣬ�÷�Ӧ�Ļ�ѧ����ʽ��__________________________________��
���𰸡� �� ��A 2 HCl SiCl4��3H2O===H2SiO3��4HCl
��������W��������Z��A��N��18��10��8����WΪ����X��Ne�ĺ�����������1����ͼ��X��ԭ�Ӱ뾶����W����XΪ�ƣ�Y�ĵ�����һ�ֳ����İ뵼����ϣ���YΪ�裻Z��ԭ����������Y������ͬ��������Ԫ���е縺�������ZΪ�ȡ�
��1��Na��Ԫ�����ڱ���λ�ڵ������ڵڢ�A�塣O�Ļ�̬ԭ�ӵĹ����ʾʽΪ![]() ������2��δ�ɶԵ��ӡ���2���ȵķǽ�����ǿ���壬��HCl���ȶ��Ը�ǿ����3��SiCl4��H2O��Ӧ����ǿ�������������ᣬ��Ӧ�Ļ�ѧ����ʽΪSiCl4��3H2O��H2SiO3��4HCl��
������2��δ�ɶԵ��ӡ���2���ȵķǽ�����ǿ���壬��HCl���ȶ��Ը�ǿ����3��SiCl4��H2O��Ӧ����ǿ�������������ᣬ��Ӧ�Ļ�ѧ����ʽΪSiCl4��3H2O��H2SiO3��4HCl��

 ��ĩ���䵥Ԫ�����ิϰ��ϵ�д�
��ĩ���䵥Ԫ�����ิϰ��ϵ�д�