��Ŀ����
����Ŀ���״�����Ϊ2l���͵�����ȼ�ϣ���ҵ�Ͽ�����CH4��H2OΪԭ�����Ʊ��״���
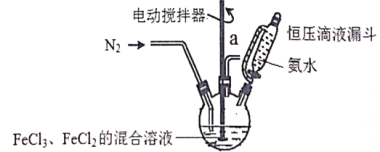
��1����1.0mol CH4��2.0mol H2O��g��ͨ���ݻ�Ϊ10L�ķ�Ӧ�ң���һ�������·�����Ӧ
��CH4��g��+H2O��g��![]() CO��g��+3H2��g����CH4��ת�������¶ȡ�ѹǿ�Ĺ�ϵ��ͼ����
CO��g��+3H2��g����CH4��ת�������¶ȡ�ѹǿ�Ĺ�ϵ��ͼ����

��֪100��,ѹǿΪP1ʱ,�ﵽƽ�������ʱ��Ϊ5min��
������ H2 ��ʾ��ƽ����Ӧ����Ϊ______��
���������������������������¶ȣ���ѧƽ�ⳣ����____����������������С����������������
��ͼ�е�P1____P2����������������������=��������100��,ѹǿΪP1ʱƽ�ⳣ��Ϊ_______��
�ܱ��ַ�Ӧ��ϵ100��,ѹǿΪP1��5min�����������г���H2O�� H2��0.5mol����ѧƽ�⽫��___�ƶ������������������һ�����������
��2����һ�������£���a mol CO�� 3a mol H2�Ļ�������ڴ������������Է�������Ӧ
��CO��g��+2H2��g��![]() CH3OH��g��
CH3OH��g��
�ٸ÷�Ӧ����H___0����S___0����������������������=������
���������ݻ����䣬���д�ʩ�������COת���ʵ���____��
A�������¶�
B����CH3OH��g������ϵ�з������
C������He��ʹ��ϵ��ѹǿ����
D���ٳ���1mol CO��3mol H2��
���𰸡�0.030molL-1min-1 ���� �� 2.25��10-2 ���� �� �� BD
��������
(1)������ͼ֪��ƽ��ʱ�����ת����,���c(CH4),����v=c/t ����v(CH4),��������֮�ȵ��ڻ�ѧ������֮�ȼ���v(H2)��
��ת�������¶ȡ�ѹǿͼ������������,Ӧ����һ�������
�����ݶ�һ���ԭ��,���¶�ͬ,�ٱȽ�ѹǿ,������ֱx��ĸ�����,�Ƚ�ƽ��ʱ�����ת����,�ɴ��ж�;ƽ�ⳣ��k=c(CO)c2(H2)/c(CH4)c(H2O)�������ƽ��ʱ����ֵ�Ũ��,����ƽ�ⳣ������ʽ������
�����ַ�Ӧ��ϵΪ100��,5min�����������г���H2O��H2��0.5mol,����Ũ������ƽ�ⳣ������Դ�С���ж���
(2)����������ڴ������������Է���Ӧ���ɼ״�,��˵������Ӧ�Ƿ��ȷ�Ӧ,����Ӧ���������ʵ�����С�ķ�Ӧ��
�������ݻ�����,���Ӽ״�����,ƽ��������Ӧ�ƶ�,�������������ƽ���Ӱ�������
(1)����100��ƽ��CH4��ת����Ϊ0.5����֪��,����CH4Ϊ1��0.5=0.5mol��ƽ��ʱ�����Ũ�ȱ仯��Ϊ:0.5/10=0.05mol/L,���ݼ����������ļ�������ϵ,��: v(H2)=3 v(CH4)=3��0.05/5=0.030molL-1min-1����ˣ�������ȷ����: 0.030molL-1min-1��
����ѹǿΪp1ʱ,�����¶�,�����ת�������,ƽ���������ƶ�,��������Ϊ���ȷ�Ӧ,���������¶�ƽ�ⳣ������������������������������
��ͨ��ͼ������֪�����¶���ͬʱ,p2��p1ʱ,�����ת�������,ƽ���������ƶ�,����Ϊ����ϵ������ķ�Ӧ,���ݼ�Сѹǿƽ����ϵ������ķ����ƶ��ɵó�p1< p2����ˣ�������ȷ����: ����
�� CH4��g��+H2O��g��![]() CO��g��+3H2��g��
CO��g��+3H2��g��
��ʼŨ��: 0.1 0.2 0 0
�仯Ũ��: 0.05 0.05 0.05 0.15
ƽ��Ũ��: 0.05 0.15 0.05 0.15
100��ʱƽ�ⳣ��=0.05����0.15��3/0.05��0.15= 2.25��10-2��5min�����������г���H2O�� H2��0.5mol��������Ũ��Ϊ��c(H2O)=0.15+0.05=0.2mol/L��c(H2)= =0.15+0.05=0.2mol/L��QC=0.23��0.05/0.05��0.2=0.04> 2.25��10-2������Ӧ����С���淴Ӧ������ƽ�������ƶ����������������������2.25��10-2������
(2)����������ڴ������������Է���Ӧ���ɼ״�,��˵������Ӧ�Ƿ��ȷ�Ӧ,������H<0 ������Ӧ���������ʵ�����С�ķ�Ӧ,��������ʵ���Խ��,����Խ��,���ԡ�S<0����ˣ�������ȷ����:<�� <��
��A.�÷�ӦΪ���ȷ�Ӧ,�����¶�,ƽ�������ȷ����ƶ�,�����淴Ӧ�����ƶ�, COת���ʽ�������A������
B.�� CH3OH��g������ϵ�з���,�����Ũ�Ƚ�����ƽ��������Ӧ�ƶ���COת������������B��ȷ��
C.����He��ʹ��ϵ��ѹǿ�����������ݻ���������Ӧ���������Ũ�Ȳ�����ƽ�ⲻ�ƶ�, COת���ʲ�������C������
D.�ٳ���1mol CO��3mol H2���ɵ�ЧΪѹǿ����,ƽ���������С�ķ����ƶ�,��������Ӧ�����ƶ���COת������������D��ȷ��
��ˣ�������ȷ����:BD��

����Ŀ���¶�ΪT1ʱ��������X������Y��1.6mol����10L�����ܱ������У�������ӦX(g)+Y(g)![]() 2Z(g)��һ��ʱ���ﵽƽ�⡣��Ӧ�����вⶨ���������������˵����ȷ����
2Z(g)��һ��ʱ���ﵽƽ�⡣��Ӧ�����вⶨ���������������˵����ȷ����
t/min | 2 | 4 | 7 | 9 |
n��Y��/mol | 1.2 | 1.1 | 1.0 | 1.0 |
A. ��Ӧ0��4 min��ƽ�����ʦ�(Z)=0.25 mol/(Lmin)
B. T1ʱ����Ӧ��ƽ�ⳣ��K1��1.2
C. �����������䣬9 min�����������ٳ���1.6 molX��ƽ��������Ӧ�����ƶ����ٴδﵽƽ��ʱX��Ũ�ȼ�С��Y��ת��������
D. �����������䣬���µ�T2�ﵽƽ��ʱ��ƽ�ⳣ��K2=4����˷�Ӧ�ġ�H��0