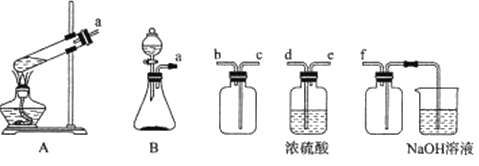
��Ŀ����
����Ŀ��ʵ����������80mL0.1mol/L��̼������Һ����ղ��ش��������⣺
��1������80mL1.0mol/L��Na2CO3��Һ��
ʵ��Ӧ��Na2CO3����/g | Ӧѡ������ƿ�Ĺ��/mL |
___ | ___ |
��2������ʱ������ȷ�IJ���˳��Ϊ___������ĸ��ʾ��ÿ����ĸֻ����һ�Σ���
A.��ȷ������Na2CO3���嵹���ձ��У��ټ�����ˮ�ܽ�
B.������ƿ�ǽ������µߵ���ҡ��
C.������ȴ����Һ�ز�����ע������ƿ��
D.��30 mLˮϴ���ձ�2��3�Σ�ϴ��Һ��ע������ƿ����
E������������ƿ��С�ļ�ˮ��ֱ��Һ��ӽ��̶���1~2cm��
F����Ϊ��ͷ�ιܼ�ˮ��ʹ��Һ��Һ��ǡ����̶�������
��3�������������������������ҺŨ�Ƚ��к�Ӱ�죿(�ƫ�ߡ���ƫ�͡�����Ӱ�족)
������ʱ���ӿ̶��ߣ������___��
������ƿ������������ˮ�������___��
���𰸡�10.6 100 ACDEFB ƫ�� ��Ӱ��
��������
(1)����������Һ���ѡ������ƿ�������m=cVM������Ҫ���ʵ�������
(2)�ù�������һ�����ʵ���Ũ����Һһ�㲽��Ϊ�����㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ����ǩ�ȣ��ݴ�����
(3)����![]() �������
�������
(1)ʵ����������80mL1.0mol/L�ĵ�̼������Һ��ʵ����û��80mL�������ƿ��Ӧѡ��100mL����ƿ����Ҫ���ʵ�����m=1molL-1��0.1L��106g/mol=10.6g���ʴ�Ϊ��10.6g��100mL��
(2)�ù�������һ�����ʵ���Ũ����Һһ�㲽��Ϊ�����㡢�������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ����ǩ�ȣ����� ��ȷ��˳��Ϊ��ACDEFB��
(3)������ʱ���ӿ̶��ߣ��ᵼ����Һ�����ƫ�����ƫ�ͣ�������ƿ������������ˮ�������ûӰ�죬�ʴ�Ϊ��ƫ�ͣ���Ӱ�졣

����Ŀ���ζ�������һ�ֲ�����㡢ȷ�ȺܸߵĶ����������������ɹ㷺Ӧ�����к͵ζ���������ԭ��Ӧ�ȵζ��С�ij�о���ѧϰС���ͬѧ���õζ��������������������������
��1���ⶨNaOH��Na2CO3�Ļ��Һ��NaOH�ĺ�����ʵ�����Ϊ������Һ�мӹ�����BaCl2��ҺʹNa2CO3��ȫת����BaCO3������Ȼ���ñ�����ζ�(�÷�̪��ָʾ��)��
�������BaCO3������NaOH��Һ��ֱ�ӵ������ᣬ���յ���ɫ�ı仯Ϊ____��Ϊ�δ�������ܲ��NaOH�ĺ�����____��
�ڵζ�ʱ�����ζ����еĵζ�Һһֱ�½����������Ŵﵽ�ζ��յ㣬���ܷ��ɴ�ȷ�ؼ���������____����˵�����ɣ�___��
��2���ⶨijƷ�Ƶĵ���(���е����)�е�Ԫ�صİٷֺ�����ȷ��ȡ5.0000g�õ��Σ���������ˮ��Ȼ����������KI��Һ�����������»��(�����ķ�ӦΪKIO3��3H2SO4��5KI��3K2SO4��3I2��3H2O)����ַ�Ӧ�����Һϡ����250 mL��Ȼ����5.0��10��4mol��L��1��Na2S2O3����Һ���еζ�(�õ�����ָʾ������ӦΪI2��2S2O32-��2I����S4O62-)��ȡ��Na2S2O3�ı���ҺӦ����_____ʽ�ζ��ܡ��й�ʵ����ֵ�����ʾ(��һ�εζ��յ��������ͼ��ʾ���뽫���õ������������)��
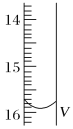
�ζ����� | ����Һ�����(mL) | �ζ�ǰ�Ķ���(mL) | �ζ���Ķ���(mL) |
��һ�� | 25.00 | 0.00 | V��_____ |
�ڶ��� | 25.00 | 0.00 | 14.99 |
������ | 25.00 | 0.00 | 15.01 |
�õ����е�Ԫ�صİٷֺ���Ϊ_____�����в����У��ᵼ������õĵ�Ԫ�صİٷֺ���ƫ�����____��
a���ζ��յ�ʱ�����ӿ̶�
b��û����Na2S2O3����Һ��ϴ��Ӧ�ĵζ���
c����ƿ��������������ˮ