��Ŀ����
��8�֣���ҵ���к���NaNO2����ۺ�ʳ�����ƣ�����ζ��������ʳ�������ж���������Ϊ0��3g��0��5g����֪NaNO2�ܷ������·�Ӧ��aNO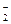 +xI��+yH+=bNO��+cI2+zH2O��
+xI��+yH+=bNO��+cI2+zH2O��
��ش��������⣺
��1��������Ӧ�У�x��y��z����ֵ�ֱ��� ��ȫ�ԲŸ��֣���
��2������������Ӧ�����û�ѧ��ֽ�������г������ʽ���ʵ��������ҵ�κ�ʳ�Σ����е⻯�ص�����ֽ������ѡ�õ������г������ʵ�����Ϊ ��
��3��ij����������Һ�к���2%��5%��NaNO2��ֱ���ŷŻ����ˮ��Ⱦ�����������������е�ijһ�־���ʹNaNO2ת��Ϊ��������Ⱦ��N2���������� ��
a��NaCl b��H2O2 c��NH4Cl d��ŨH2SO4
��������Ӧ�����ӷ���ʽΪ�� ��
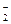 +xI��+yH+=bNO��+cI2+zH2O��
+xI��+yH+=bNO��+cI2+zH2O����ش��������⣺
��1��������Ӧ�У�x��y��z����ֵ�ֱ��� ��ȫ�ԲŸ��֣���
��2������������Ӧ�����û�ѧ��ֽ�������г������ʽ���ʵ��������ҵ�κ�ʳ�Σ����е⻯�ص�����ֽ������ѡ�õ������г������ʵ�����Ϊ ��
��3��ij����������Һ�к���2%��5%��NaNO2��ֱ���ŷŻ����ˮ��Ⱦ�����������������е�ijһ�־���ʹNaNO2ת��Ϊ��������Ⱦ��N2���������� ��
a��NaCl b��H2O2 c��NH4Cl d��ŨH2SO4
��������Ӧ�����ӷ���ʽΪ�� ��
��8�֣�
��1��2�� 4�� 2
��2��ʳ��
��3��c NH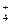 +NO
+NO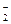 =N2��+2H2O
=N2��+2H2O
��1��2�� 4�� 2
��2��ʳ��
��3��c NH
 +NO
+NO =N2��+2H2O
=N2��+2H2O��

��ϰ��ϵ�д�
�����Ŀ
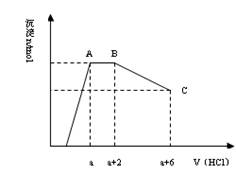


 ��HCO3��2��Һ�е���NaHSO4��Һ����������ȫ����Ӧ�����ӷ���ʽ_________________________��������Һ�м�������NaHSO4��Һ����ʱ���ӷ���ʽΪ_______________________��
��HCO3��2��Һ�е���NaHSO4��Һ����������ȫ����Ӧ�����ӷ���ʽ_________________________��������Һ�м�������NaHSO4��Һ����ʱ���ӷ���ʽΪ_______________________�� O4 =" Cu" + FeSO4
O4 =" Cu" + FeSO4 2 NaCl
2 NaCl 2KCl + 3O2��
2KCl + 3O2�� ���е������� ���������ʾ�д��ѧʽ����д���ڢٲ��з�Ӧ�Ļ�ѧ����ʽ��
���е������� ���������ʾ�д��ѧʽ����д���ڢٲ��з�Ӧ�Ļ�ѧ����ʽ��  ��
��