��Ŀ����
����Ŀ��ҳ�����к��н϶�����飬�ɽ���ת��Ϊ���й�ҵ��ֵ����ϩ��
��1�� ������̼������������ϩ��
��C2H6��CO2�����ʵ���֮��Ϊ1��1ͨ�뷴Ӧ���У��������·�Ӧ��
����C2H6(g) ![]() C2H4(g) + H2(g) ��H1��+136.4 kJ��mol 1
C2H4(g) + H2(g) ��H1��+136.4 kJ��mol 1
����CO2(g) + H2(g) ![]() CO(g) + H2O(g) ��H2��+41.2 kJ��mol 1
CO(g) + H2O(g) ��H2��+41.2 kJ��mol 1
����C2H6(g) +CO2(g) ![]() C2H4(g) +CO(g) +H2O(g) ��H3
C2H4(g) +CO(g) +H2O(g) ��H3
������H1����H2������H3��______kJ��mol1��
�ڷ�Ӧ����C2H6(g) ![]() 2C(s)+3H2(g)Ϊ��̼��Ӧ�����ɵ�̼�����ڴ������棬 ���ʹ����Ļ��ԣ��ʵ�ͨ����� CO2 ������Ч�����̼����Ϸ���ʽ������ԭ��__��
2C(s)+3H2(g)Ϊ��̼��Ӧ�����ɵ�̼�����ڴ������棬 ���ʹ����Ļ��ԣ��ʵ�ͨ����� CO2 ������Ч�����̼����Ϸ���ʽ������ԭ��__��
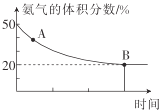
�۶�����̼������������ϩ���о��ȵ�֮һ��ѡ���������ͬ��Ӧʱ�䣬��ͬ�¶ȡ���ͬ�������������±�����δ�ﵽƽ��״̬����

��ע��C2H4 ѡ���ԣ�ת����������������ϩ�İٷֱȡ�
CO ѡ���ԣ�ת���� CO2 ������ CO �İٷֱȡ�
�ԱȢ�͢÷�ӦӦ��ѡ��Ĵ���Ϊ__��������__��ʵ�������£�����������ʱ�����¶����ߣ�C2H6 ��ת�������ߣ��� C2H4 ��ѡ���Խ��ͣ�ԭ����__��
��2�� �������Ӵ���������������ؽ�����ת��Ϊ��ϩ��ʾ��ͼ��ͼ��
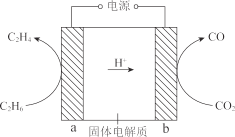
�ٵ缫 a ���Դ��______��������
�ڵ缫 b �ĵ缫��Ӧʽ�� ______��
���𰸡�+177.6 ����CO2������������ӦC+CO2 ![]() 2CO������C������CO2��������
2CO������C������CO2��������
Ӧ��������г̶����ӣ�������C2H6��Ũ�ȣ���Ӧ�����еij̶ȼ�С ���� ��ͬ�¶��£�����������ʱC2H6��ת���ʺ�C2H4��ѡ���Ծ��ϸ� �¶����ߣ���Ӧ�����������Ļ�ѧ��Ӧ���ʾ�����Ӧ������ĸ��� ���� CO2 +2e��+2H+=CO+H2O
��������
��1������֪��C2H6(g) ![]() C2H4(g) + H2(g) ��H1��+136.4 kJ��mol 1
C2H4(g) + H2(g) ��H1��+136.4 kJ��mol 1
����CO2(g) + H2(g) ![]() CO(g) + H2O(g) ��H2��+41.2 kJ��mol 1
CO(g) + H2O(g) ��H2��+41.2 kJ��mol 1
����C2H6(g) +CO2(g) ![]() C2H4(g) +CO(g) +H2O(g) ��H3
C2H4(g) +CO(g) +H2O(g) ��H3
�ɸ�˹���ɿ�֪����Ӧ��+��Ӧ���÷�Ӧ��������H3����H1+��H2=��+136.4 kJ��mol 1��+��+41.2 kJ��mol 1��=+177.6 kJ��mol 1���ʴ�Ϊ��+177.6��
�����������̼������������Ч�����̼��ԭ���ǣ�������̼����̼�ڼ��������·�Ӧ����һ����̼�����Ļ�̼����Ӧ�Ļ�ѧ����ʽΪC+CO2 ![]() 2CO�����������̼��������Ӧ����������Ӧ�����ƶ�����������ϵ��C2H6��Ũ�ȣ�ʹ��Ӧ�����淴Ӧ�����ƶ������ٻ�̼�����ɣ��ʴ�Ϊ������CO2������������ӦC+CO2
2CO�����������̼��������Ӧ����������Ӧ�����ƶ�����������ϵ��C2H6��Ũ�ȣ�ʹ��Ӧ�����淴Ӧ�����ƶ������ٻ�̼�����ɣ��ʴ�Ϊ������CO2������������ӦC+CO2 ![]() 2CO������C������CO2��������Ӧ��������г̶����ӣ�������C2H6��Ũ�ȣ���Ӧ�����еij̶ȼ�С��
2CO������C������CO2��������Ӧ��������г̶����ӣ�������C2H6��Ũ�ȣ���Ӧ�����еij̶ȼ�С��
���ɱ������ݿ�֪����ͬ�¶��£�����������ʱ��C2H6��ת���ʺ�C2H4��ѡ���Ծ��ϸߣ���ԱȢ�͢÷�ӦӦ��ѡ��Ĵ���Ϊ���Σ������¶ȣ���Ӧ�����������Ļ�ѧ��Ӧ���ʾ�����Ӧ���Ļ�ѧ��Ӧ��������ķ��ȸ�����C2H6 ��ת�������ߣ�C2H4��ѡ���Խ��ͣ��ʴ�Ϊ���¶����ߣ���Ӧ�����������Ļ�ѧ��Ӧ���ʾ�����Ӧ������ĸ��ࣻ
��2���ٵ����У��������������ƶ�����ͼ�������ӵ��ƶ������֪���缫 aΪ���������Դ�����������ʴ�Ϊ������
�ڵ����У��������������ƶ�����ͼ�������ӵ��ƶ������֪���缫bΪ�������������������£�������̼�������ϵõ��ӷ�����ԭ��Ӧ����һ����̼��ˮ���缫��ӦʽΪCO2 +2e��+2H+=CO+H2O��

 �Ͻ�ƽСѧ��������ϵ�д�
�Ͻ�ƽСѧ��������ϵ�д�����Ŀ�����������Դ�ǵ����о���һ���ȵ����⡣�����ѣ�CH3OCH3����δ������������ͺ�Һ������Ϊ�ྻҺ��ȼ��ʹ�ã���ҵ����CO��H2Ϊԭ������CH3OCH3����ҵ�Ʊ��������ڴ���Ӧ���У�ѹ��2.0��10.0Mpa���¶�230��280�棩�������з�Ӧ��
��CO(g)��2H2(g)![]() CH3OH(g) ��H1=-99kJ��mol-1
CH3OH(g) ��H1=-99kJ��mol-1
��2CH3OH(g)![]() CH3OCH3(g)��H2O(g) ��H2=-23.5kJ��mol-1
CH3OCH3(g)��H2O(g) ��H2=-23.5kJ��mol-1
��CO(g)��H2O(g)![]() CO2(g)��H2(g) ��H3=-41.2kJ��mol-1
CO2(g)��H2(g) ��H3=-41.2kJ��mol-1
��1������Ӧ���е��ܷ�Ӧ3CO(g)��3H2(g)![]() CH3OCH3(g)��CO2(g)��������H=___����Ӧԭ������ú����������֪�÷�Ӧ��ƽ�ⳣ������ʽΪK=
CH3OCH3(g)��CO2(g)��������H=___����Ӧԭ������ú����������֪�÷�Ӧ��ƽ�ⳣ������ʽΪK=![]() ��ÿ����1molH2��Ҫ����131.3kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ___��
��ÿ����1molH2��Ҫ����131.3kJ��������д���÷�Ӧ���Ȼ�ѧ����ʽ___��
��2���ڸ������£�����Ӧ�ٵ���ʼŨ�ȷֱ�Ϊ��c(CO)=0.6mol��L-1��c(H2)=1.4mol��L-1��8min��ﵽƽ�⣬CO��ת����Ϊ50%����8min��H2��ƽ����Ӧ����Ϊ___��
��3����t��ʱ����Ӧ�ڵ�ƽ�ⳣ��Ϊ400�����¶��£���1L���ܱ������м���һ���ļ״�����Ӧ��ijʱ�̲�ø���ֵ����ʵ���Ũ�����£�
���� | CH3OH | CH3OCH3 | H2O |
c(mol��L��1) | 0.46 | 1.0 | 1.0 |
��ʱ��v��___v������������������������������ƽ��ʱc(CH3OCH3)�����ʵ���Ũ����___��
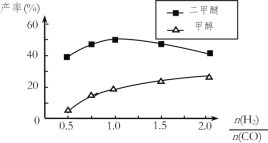
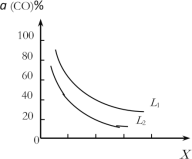
��4���ڣ�1��С���д���Ӧ�ҵ��ܷ�Ӧ3CO(g)��3H2(g)![]() CH3OCH3(g)��CO2(g)��CO��ƽ��ת����a(CO)���¶ȡ�ѹǿ�Ĺ�ϵ��ͼ��ʾ��ͼ��X����___�������¶�������ѹǿ��������L1___L2��������������������������
CH3OCH3(g)��CO2(g)��CO��ƽ��ת����a(CO)���¶ȡ�ѹǿ�Ĺ�ϵ��ͼ��ʾ��ͼ��X����___�������¶�������ѹǿ��������L1___L2��������������������������
��5���ڴ�����������ͬʱ����������Ӧ������������ʼͶ�ϱ�![]() �ĸı�����Ѻͼ״��IJ��ʣ������е�̼ԭ��ռ��ʼCO��̼ԭ�ӵİٷ��ʣ�������ͼ�ı仯���ơ��Խ���Ͷ�ϱȴ���1.0֮������Ѳ��ʺͼ״����ʱ仯��ԭ��__��
�ĸı�����Ѻͼ״��IJ��ʣ������е�̼ԭ��ռ��ʼCO��̼ԭ�ӵİٷ��ʣ�������ͼ�ı仯���ơ��Խ���Ͷ�ϱȴ���1.0֮������Ѳ��ʺͼ״����ʱ仯��ԭ��__��