题目内容
【题目】某化学实验小组同学利用以下装置制备氨气,并探究氨气的性质(部分仪器已略去)。请回答下列问题:
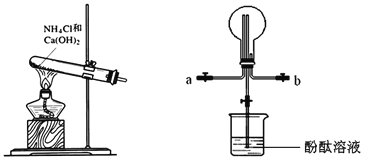
A B
(1)实验室制备氨气的化学方程式为______;干燥氨气常用的干燥剂是_____。
(2)收集氨气时,请你选择氨气的进气口_________(填“a”或“b”)。
(3)①、若观察到装置B中的烧瓶内产生了红色喷泉,则说明氨气具有的性质是_____。
②、若装置B中的烧瓶充满干燥气体,烧杯中盛有液体,下列组合中不可能形成喷泉的是___
A HCl和H2O B H2和H2O C CO2和NaOH
(4)氨气在催化剂并且加热时会被空气氧化,这是工业制硝酸的第一步反应,写出该反应的化学方程式__________________________________。
【答案】(1)2NH4Cl+Ca(OH)2==2NH3↑+2H2O+CaCl2(2分) 碱石灰 (1分)
(2)a (1分)
(3)①NH3极易溶于水且水溶液呈碱性(2分) ②B(2分)
(4)4NH3+5O2![]() 4NO+6H2O(2分)
4NO+6H2O(2分)
【解析】
试题:(1)实验室用加热氯化氨和消石灰的固体混合物制取氨气,故答案为:2NH4Cl+Ca(OH)2![]() 2NH3↑+2H2O+CaCl2;得到的氨气为碱性气体,可以用碱石灰(碱性干燥剂,氢氧化钠固体和氧化钙的混合物)干燥。
2NH3↑+2H2O+CaCl2;得到的氨气为碱性气体,可以用碱石灰(碱性干燥剂,氢氧化钠固体和氧化钙的混合物)干燥。
(2)氨气的式量为17,空气的平均式量为29,氨气密度小于空气的密度,所以要采用向下排空气法收集氨气,即a 为进气口,b为出气口,故答案为:a;
(3)①、氨气溶于水生成氨水,氨水电离出自由移动的氢氧根离子,所以溶液呈碱性,加入酚酞后溶液呈红色;
②、氨气形成喷泉的原理是由于氨气极易溶于水,氨气溶于水而导致烧瓶内压强减小,从而形成喷泉。依据氨气形成喷泉的原理,如要形成喷泉,烧瓶内气体溶于水导致烧瓶内压强减小,外界大气压将烧杯内液体压入到烧瓶内,从而形成喷泉;因为H2不溶于水,不能形成喷泉,所以本题选B。
(4)氨催化氧化法制硝酸的步骤:氨气催化氧化为一氧化氮、一氧化氮和氧气反应生成二
氧化氮、二氧化氮和水反应生成硝酸,故答案为:4NH3+5O2![]() 4NO+6H2O。
4NO+6H2O。






