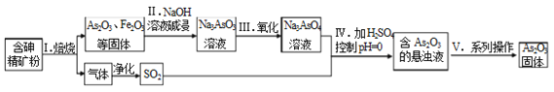
��Ŀ����
����Ŀ���£�����ʽ N2H4���е� 113.5�棩��һ�����õĻ��ȼ�ϣ���һ������ ��ˮ�ʹ�����л��ܼ�����ɫ��״Һ�塣�ڹ�ҵ������Ҫ��;��ʵ���ҿ������� װ�ã���ȥ���ּг�װ�ã�ģ�����ð����ʹ������ƺϳ��µĹ��̡�

��1��װ�� A �������Ʊ� Cl2��װ�� B �б���ʳ��ˮ��������_____�����ϳ��µķ�Ӧ����ʱ�ر�ֹˮ�� K1��װ�� B ���Թ۲쵽��ʵ������_____��
��2��װ�� C �г��������� b ��װ�� B �е�_____�����������ƣ���������ͬ��
��3��װ�� E �п�����ʯ�Һ��Ȼ���Ʊ� NH3����Ӧ�Ļ�ѧ����ʽΪ_____
��4������Ӧһ��ʱ�䣬�ر�����ֹˮ�У���װ�� C �з�Һ©���Ļ���������Һ ȫ�����뼯��ƿ�У���Ӧ�����¡���Ӧ�����ӷ���ʽΪ_____�� ��ͬѧ˵װ�� D û��ʲô���ã���ȥ��װ�� D����ʵ���Ƿ���Ӱ�죿��˵�����ɣ�___________��
��5���������� Cl2 �뺬�� 0.4mol NaOH ��ˮ��Һ��Ӧ����Һ©����Һ��ȫ������ C �У����� C ��ͨ���״���µ� 13.44L NH3���� C ���������õ� 3.6g Һ̬�£� ���ʵ�����µ�ʵ�����Ϊ___��
���𰸡���ȥCl2�е�HCl���� ����ƿ��Һ���½����ϲ���������ɫ���壬����©���е�Һ������ ����©��a Ca(OH)2+2NH4Cl![]() 2NH3��+CaCl2+2H2O 2NH3H2O +ClO- = N2H4+Cl-+3H2O ��Ӱ�죬NH3����ˮ��C��Һ��������Eװ���Թ��У������Թ�ը�� 56.25%
2NH3��+CaCl2+2H2O 2NH3H2O +ClO- = N2H4+Cl-+3H2O ��Ӱ�죬NH3����ˮ��C��Һ��������Eװ���Թ��У������Թ�ը�� 56.25%
��������
��1��װ�� A�Ʊ��� Cl2���������Ȼ��⣬���ϳ��µķ�Ӧ����ʱ�ر�ֹˮ�� K1��Bװ���ռ������������
��2��װ�� C �г��������� b ��װ�� B �еij���©��������ƽ����ѹ�����á�
��3��װ�� E �м�����ʯ�Һ��Ȼ�立�Ӧ����NH3���Ȼ��ơ�ˮ��
��4������Ӧһ��ʱ�䣬C �з�Һ©�������ɴ������ƣ�����Һȫ�����뼯��ƿ�У��������������Ʒ�Ӧ�����¡��Ȼ��ơ�ˮ�� ���ݰ���������ˮ����װ��D�����á�
��5����Һ©���еķ�Ӧ����ʽ��2NaOH+Cl2=NaCl+NaClO+H2O��C�з�Ӧ���ӷ���ʽ��2NH3H2O +ClO- = N2H4+Cl-+3H2O���ɵù�ϵʽ2NaOH��2NH3H2O�� N2H4�����ݹ�ϵʽ�����µIJ��ʡ�
��1��װ�� A�Ʊ��� Cl2���������Ȼ��⣬װ�� B �б���ʳ��ˮ�������dz�ȥCl2�е�HCl���ʣ����ϳ��µķ�Ӧ����ʱ�ر�ֹˮ�� K1��Bװ���ռ�����������������Ǽ���ƿ��Һ���½����ϲ���������ɫ���壬����©���е�Һ��������
��2��װ�� C �г��������� b ��װ�� B �еij���©��������ƽ����ѹ�����ã�����װ�� C �г��������� b ��װ�� B �еij���©��a��������ͬ��
��3��װ�� E �м�����ʯ�Һ��Ȼ������NH3���Ȼ��ơ�ˮ����Ӧ�Ļ�ѧ����ʽ��Ca(OH)2+2NH4Cl![]() 2NH3��+CaCl2+2H2O��
2NH3��+CaCl2+2H2O��
��4������Ӧһ��ʱ�䣬C �з�Һ©�������ɴ������ƣ�����Һȫ�����뼯��ƿ�У��������������Ʒ�Ӧ�����¡��Ȼ��ơ�ˮ����Ӧ���ӷ���ʽ��2NH3H2O +ClO- = N2H4+Cl-+3H2O������������ˮ����ȥ��װ�� D ��C��Һ��������Eװ���Թ��У������Թ�ը�ѣ�������Ӱ�졣
��5��C��Һ©���еķ�Ӧ����ʽ��2NaOH+Cl2=NaCl+NaClO+H2O��C �з�Ӧ���ӷ���ʽ��2NH3H2O +ClO- = N2H4+Cl-+3H2O���ɵù�ϵʽ2NaOH��2NH3H2O�� N2H4�����ݹ�ϵʽ��0.4mol NaOH����������0.2mol N2H4���µIJ�����![]() 56.25% ��
56.25% ��

 ������ӱ������ͯ������ϵ�д�
������ӱ������ͯ������ϵ�д� A�ӽ��� ϵ�д�
A�ӽ��� ϵ�д� ȫ�Ų��Ծ�ϵ�д�
ȫ�Ų��Ծ�ϵ�д�����Ŀ��������±�ᷴӦ���Ʊ�±��������Ҫ������ʵ�����Ʊ��������1-�嶡��ķ�Ӧ���£�
NaBr+H2SO4=HBr+NaHSO4 ��
R-OH+HBr![]() R-Br+H2O ��
R-Br+H2O ��
���ܴ��ڵĸ���Ӧ�У�����Ũ����Ĵ�������ˮ����ϩ���ѣ�Br-��Ũ��������ΪBr2�ȡ��й������б����£�
�Ҵ� | ������ | ������ | 1-�嶡�� | |
�ܶ�/g��cm-3 | 0.7893 | 1.4604 | 0.8098 | 1.2758 |
�е�/�� | 78.5 | 38.4 | 117.2 | 101.6 |
��ش�
��1���õ����������к��������Ҵ���Ϊ���Ƶô����������飬��������ˮϴ�ӣ���Һ���ټ�����ˮCaCl2��Ȼ����е�ʵ�������___������ĸ����
a����Һ b������ c����ȡ d������
��2���������ˮ����___�Ҵ���������������������������С����������ԭ����___��
��3����1-�嶡��ֲ�Ʒ���ڷ�Һ©���м�ˮ�����ã�������___�������ϲ��������²����������ֲ�������
��4���Ʊ������У������Ũ����������ϡ�ͣ���Ŀ����___������ĸ����
a�����ٸ�����ϩ���ѵ����� b������Br2������
c������HBr�Ļӷ� d��ˮ�Ƿ�Ӧ�Ĵ���
��5��Ϊ�˼����������к�����Ԫ�أ�ͨ�����õķ����ǣ�ȡ���������飬Ȼ��������в������ټ��� �ڼ���AgNO3��Һ �ۼ���ϡHNO3�ữ �ܼ���NaOH��Һ�����в���˳���������___������ĸ����
a���٢ڢۢ� b���ڢۢ٢� c���ܢ٢ۢ� d�� �٢ܢڢ�
��6�����Ʊ�������ʱ�����ñ߷�Ӧ����������ķ�������������___��
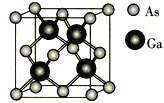
����Ŀ��GaN������5GоƬ�IJ��ϣ����������͵�����LED�ɷ�������⡣�ش��������⣺
(1)��̬Asԭ�Ӻ�������Ų�ʽΪ[Ar]________������״̬�����У�����������һ����������������С����_____________(����)��
A. ![]() B.
B. ![]() C.[Ne] D.
C.[Ne] D. ![]()
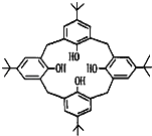
(2)8���ǻ������(����ʽC27H18AlN3O3)���ڷ�����ϼ����Ӵ�����ϣ�����LiAlH4�� (8���ǻ����)�ϳɡ�LiAlH4�������ӵĿռ乹��Ϊ_______��
(8���ǻ����)�ϳɡ�LiAlH4�������ӵĿռ乹��Ϊ_______�� ����Ԫ���е縺��������___(��Ԫ�ط���)��C��N��O���ӻ���ʽ����Ϊ _____��_________��____________��
����Ԫ���е縺��������___(��Ԫ�ط���)��C��N��O���ӻ���ʽ����Ϊ _____��_________��____________��
(3)��֪���л�������۵㣺
������ | AlF3 | GaF3 | AlCl3 |
�۵�/�� | 1040 | 1000 | 194 |
�ٱ���±������۵���������ԭ����____________��
������AlCl3ʱ�����ɾ��лӷ��ԵĶ�����Al2Cl6��������Al2Cl6�ĽṹʽΪ_______������Al����λ��Ϊ_________��
(4)GaAs�ľ����ṹ��ͼ��ʾ�����ڵ�Asԭ��֮��ľ���Ϊx,���ڵ�As��Gaԭ��֮��ľ���Ϊy����![]() =________��
=________��
����Ŀ��1Lij�����Һ��ֻ���ܺ����±��е����������ӣ�
���ܴ������е������� |
|
���ܴ������е������� |
|
�������Һ�м���һ�����ʵ���Ũ�ȵ�NaOH��Һʱ���������ɳ��������ʵ�����NaOH��Һ������仯��ͼ����ͼ��ʾ������˵����ȷ����![]()
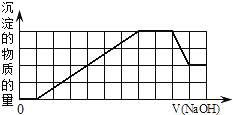
A.ԭ��Һ�м��������г�������
B.ԭ��Һ��һ������![]() ��
��![]()
C.��Ӧ���õ��ij���ֻ��![]()
D.��Ӧ����γɵ���Һ������ֻ��![]()