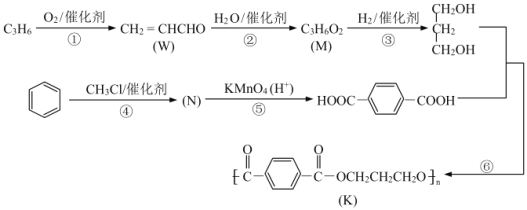
��Ŀ����
����Ŀ�������и�����Һ���������ӵ��жϺ������ǣ�������
A.����ɫ��Һ�м���ˮ���ɫ����Һ�п��ܺ���SO42-��Br-��OH-��Ba2+
B.25��ʱ��ˮ�������c(H+)=1.0��10-11mol/L����Һ�п��ܺ���Mg2+��Cu2+��SO42-��NO3-
C.25��ʱ![]() ��0.1 mol/L����Һ�п��ܺ���Na����K����CO32-��NO3-
��0.1 mol/L����Һ�п��ܺ���Na����K����CO32-��NO3-
D.��c(Fe3+)=1.0mol/L����Һ�п��ܺ���K+��Na+��SCN-��HCO3-
���𰸡�B
��������
A. ����ɫ��Һ�м���ˮ���ɫ����Һ��һ����Br-����һ������OH-������ͬʱ��SO42-��Ba2+��A����
B. 25����ˮ�������c(H+)=1.0��10-11mol/L����Һ�������Ǽ���Һ��Ҳ����������Һ������ҺΪ����Һ������ܺ���Mg2+��Cu2+��SO42-��NO3-��B��ȷ��
C. c(H+)=![]() ��0.1 mol/L������Һ�����ԣ��ڴ���Һ��CO32-���ܴ������ڣ�C����
��0.1 mol/L������Һ�����ԣ��ڴ���Һ��CO32-���ܴ������ڣ�C����
D. ��c(Fe3+)=1.0mol/L����Һ�У������ܺ�SCN-��HCO3-��D����
��ѡB��

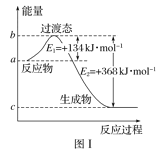
����Ŀ���ο�����ͼ�����й�Ҫ��ش����⣺
��1��ͼ����1molNO2(g)��1molCO(g)��Ӧ����CO2��NO�����������仯ʾ��ͼ�����ڷ�Ӧ��ϵ�м����������Ӧ��������E1�ı仯��___(��������������С����������������ͬ)����H�ı仯��__����д��NO2��CO��Ӧ���Ȼ�ѧ����ʽ��__��
��2���״����ӽ���Ĥȼ�ϵ���н��״�����ת��Ϊ���������ַ�Ӧԭ���ǣ�
��CH3OH(g)��H2O(g)=CO2(g)��3H2(g) ��H����49.0kJ��mol��1
��CH3OH(g)��![]() O2(g)=CO2(g)��2H2(g) ��H����192.9kJ��mol��1
O2(g)=CO2(g)��2H2(g) ��H����192.9kJ��mol��1
��֪��H2O(g)=H2O(l) ��H����44kJ��mol��1
��״�ȼ������Һ̬ˮ���Ȼ�ѧ����ʽ��___��
��3���±��Dz��ֻ�ѧ���ļ������ݣ�
��ѧ�� | P��P | P��O | O=O | P=O |
����/kJ��mol��1 | a | b | c | x |
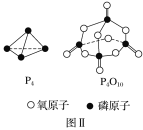
��֪1mol����(P4)��ȫȼ�շ���ΪdkJ����������ȫȼ�յIJ���ṹ��ͼ����ʾ�������x��___kJ��mol��1(�ú���a��b��c��d�Ĵ���ʽ��ʾ)��
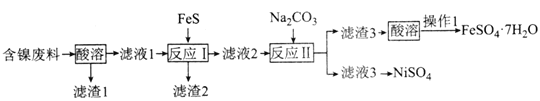
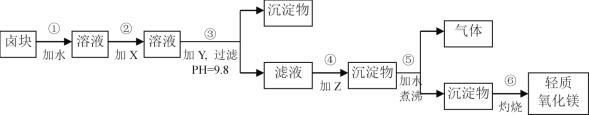
����Ŀ��±�����Ҫ�ɷ���MgCl2�������Fe3+��Fe2+��Mn2+�����ӡ�������Ϊԭ�ϰ���ͼ��ʾ�������̽������������Ƶ���������þ��
��1
���� | ��ʼ���� | ������ȫ |
Fe��OH��3 | 2.7 | 3.7 |
Fe��OH��2 | 7.6 | 9.6 |
Mn��OH��2 | 8.3 | 9.8 |
Mg��OH��2 | 9.6 | 11.1 |
�~Fe2+�����������״�����״���Һ�г�ȥ�����뽫����������Fe(OH)3������ʽ���ܳ���
��2
���� | �۸�Ԫ/�֣� |
Ư��Һ����25.2%NaClO�� | 450 |
˫��ˮ����30%H2O2�� | 2400 |
�ռ��98%NaOH�� | 2100 |
�����99.5%Na2CO3�� | 600 |
��Ҫ���Ʒ�����������ʣ����������ɱ��ϵͣ����ݱ�1�ͱ�2�ṩ�����ϣ��ش��������⣺
(1)������м�����Լ�X�����ѡ��_____________�����������ƣ�����������___________________��
(2)��������ɵij����ﺬ��____________________(�ѧʽ)��
(3)������з�����Ӧ�Ļ�ѧ����ʽΪ_____________________________��
(4)����ʵ���ҽ��в��������ʵ�飬�����õ��������������š��ƾ���ơ�________�������ǡ�
(5)��ʵ�����У�Ϊ�˳�ȥMgCl2������Һ�е�Fe3+���õ��ϴ�����MgCl2��Һ�����ڼ��Ƚ���������¼���һ���Լ������˺�������Һ�м������������ᣬ�����Լ���____________(����ĸ)��
A��NH3��H2O B��NaOH C��Na2CO3 D��MgCO3
����Ŀ����CH3CH2CH2Br�Ʊ�CH3CH(OH)CH2OH�����η����ķ�Ӧ���ͺͷ�Ӧ��������ȷ����
ѡ�� | ��Ӧ���� | ��Ӧ���� |
A | �ӳɡ�ȡ������ȥ | KOH����Һ/���ȡ�KOHˮ��Һ/���ȡ����� |
B | ��ȥ���ӳɡ�ȡ�� | NaOH����Һ/���ȡ����¡�KOHˮ��Һ/���� |
C | ������ȡ������ȥ | ���ȡ�KOH����Һ/���ȡ�KOHˮ��Һ/���� |
D | ��ȥ���ӳɡ�ˮ�� | NaOHˮ��Һ/���ȡ����¡�NaOH����Һ/���� |
A. A B. B C. C D. D