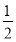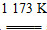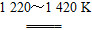��Ŀ����
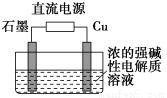
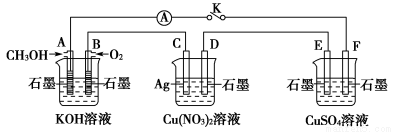
ij��ȤС���ͬѧ����ͼ��ʾװ���о��йص绯ѧ������(�ס��ҡ�����������������)�����պϸ�װ�õĿ���Kʱ���۲쵽�����Ƶ�ָ�뷢����ƫת��
�������� ���׳ء������������ҳء���������������
��ش��������⣺
(1)�ס��ҡ���������Ϊԭ��ص���________(�����׳������ҳ�������������)��
(2)������F�缫Ϊ________(������������������������������������)���óص��ܷ�ӦʽΪ__________________________________________________��
(3)���ҳ���C�缫��������10.8 gʱ���׳���B�缫����������O2�����Ϊ________mL(��״��)��
(4)һ��ʱ��Ͽ�����K������������ʹ���ػָ�����ӦǰŨ�ȵ���________(��ѡ����ĸ)��
A��Cu������������B��CuO C��CuCO3 D��Cu2(OH)2CO3
��(1)�׳ء�(2)������2CuSO4��2H2O  2H2SO4��2Cu��O2����(3)560��(4)BC
2H2SO4��2Cu��O2����(3)560��(4)BC
����������(1)�ס��ҡ���������ֻ�м׳��е�������ԭ��Ӧ���Է����У���˼׳�Ϊԭ��ء�(2)������F�缫��׳ظ���������Ϊ����������Ϊ�������ͭ��Һ���ܷ�ӦʽΪ2CuSO4��2H2O  2H2SO4��2Cu��O2����(3)�ҳ���C�缫��ӦʽΪAg��e��=Ag�����׳���B�缫��ӦʽΪ2H2O��O2��4e��=4OH�������й�ϵʽ��4 Ag��O2����V(O2)��
2H2SO4��2Cu��O2����(3)�ҳ���C�缫��ӦʽΪAg��e��=Ag�����׳���B�缫��ӦʽΪ2H2O��O2��4e��=4OH�������й�ϵʽ��4 Ag��O2����V(O2)��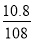 ��
��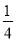 ��22.4��0.56 L��(4)�ӱ��صĵ���ܷ�Ӧʽ���Կ�������Һ�м��ٵ���2Cu��O2���൱��CuO����˿��Լ���CuO��CuCO3��
��22.4��0.56 L��(4)�ӱ��صĵ���ܷ�Ӧʽ���Կ�������Һ�м��ٵ���2Cu��O2���൱��CuO����˿��Լ���CuO��CuCO3��

 ������������Ծ�ϵ�д�
������������Ծ�ϵ�д� �������Ӳ�ϵ�д�
�������Ӳ�ϵ�д�