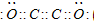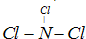��Ŀ����
����A��B��C��D��E����ǿ����ʣ�������ˮ�пɵ�������������ӣ��������Ӳ��ظ�����
��֪����A��B����Һ�ʼ��ԣ�C��D��E��Һ�����ԣ�
��A��Һ��E��Һ��Ӧ�����������г���������A��Һ��C��Һ��Ӧֻ������������������������ͬ����
��D��Һ������������Һ��Ӧ���ܲ���������Cֻ����D��Ӧ����������
�Իش��������⣺
��1����C��Һ��μ��������������ʵ�����Ũ�ȵ�A��Һ�У���Ӧ����Һ�и�������Ũ���ɴ�С��˳��Ϊ��
��2��д��E��Һ�������B��Һ��Ӧ�����ӷ���ʽ��
��3����֪��NaOH��aq��+HNO3��aq��=NaNO3��aq��+H2O��l������H=-a KJ?mol-1��
��д��B��C��ϡ��Һ��Ӧ���Ȼ�ѧ����ʽ
��4����100mL 0.1mol?L-1E��Һ�У���μ���35mL 2mol?L-1NaOH��Һ�����յõ��������ʵ���Ϊ
| ������ | H+��Na+��Al3+��Ag+��Ba2+ |
| ������ | OH-��Cl-��CO32-��NO3-��SO42- |
��A��Һ��E��Һ��Ӧ�����������г���������A��Һ��C��Һ��Ӧֻ������������������������ͬ����
��D��Һ������������Һ��Ӧ���ܲ���������Cֻ����D��Ӧ����������
�Իش��������⣺
��1����C��Һ��μ��������������ʵ�����Ũ�ȵ�A��Һ�У���Ӧ����Һ�и�������Ũ���ɴ�С��˳��Ϊ��
c��Na+����c��C1-����c��HCO3-����c��OH-����c��H+����c��CO32-��
c��Na+����c��C1-����c��HCO3-����c��OH-����c��H+����c��CO32-��
����2��д��E��Һ�������B��Һ��Ӧ�����ӷ���ʽ��
2Al3++3SO42-+3Ba2++8OH-�T2AlO2-+4H2O+3BaSO4��
2Al3++3SO42-+3Ba2++8OH-�T2AlO2-+4H2O+3BaSO4��
����3����֪��NaOH��aq��+HNO3��aq��=NaNO3��aq��+H2O��l������H=-a KJ?mol-1��
��д��B��C��ϡ��Һ��Ӧ���Ȼ�ѧ����ʽ
OH-��aq��+H+��aq��=H2O��1����H=-aKJ/mol��
Ba��OH��2��aq��+2HC1��aq��=
BaC12��aq��+H2O��1����H=-aKJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+H2O��1����H=-2aKJ/mol
| 1 |
| 2 |
| 1 |
| 2 |
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+H2O��1����H=-2aKJ/mol
OH-��aq��+H+��aq��=H2O��1����H=-aKJ/mol��
Ba��OH��2��aq��+2HC1��aq��=
BaC12��aq��+H2O��1����H=-aKJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+H2O��1����H=-2aKJ/mol
��| 1 |
| 2 |
| 1 |
| 2 |
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+H2O��1����H=-2aKJ/mol
��4����100mL 0.1mol?L-1E��Һ�У���μ���35mL 2mol?L-1NaOH��Һ�����յõ��������ʵ���Ϊ
0.01mol
0.01mol
����������A��B��Һ�ʼ��ԣ��������ĸ��������У�̼���ˮ���Լ��ԣ��������м��ԣ�����A��B�к��е�������Ϊ̼�����������������Ϊ�����Ӻ�������ˮ�������ԣ������������ԣ�����C��D��E��Һ���е�������ΪH+��Al3+��Ag+��Na2CO3�Ϳ����Ե����η�Ӧ��ʵ���ǣ������Ӻ�̼�������֮��˫ˮ�ⷴӦ��������г����������壬����A��̼���ƣ�B������������̼���ƺ��ᷴӦ�����������̼���壬�������е������Ӻ���������ɵ������У��������ܺ����ᡢ�Ȼ��ơ�������������������Ӧ���ɳ���������D����������C�����ᣬEΪ��������
��1��C��Һ��μ��������������ʵ�����Ũ�ȵ�A��Һ���У�HCl��̼���Ʒ�Ӧ����NaHCO3������ˮ�����Ƚ�����Ũ�ȵĴ�С��
��2������������������������Ӧ��������ȫ����Ӧ�������Ӻ������֮�䰴�����ʵ���֮����2��3���з�Ӧ��
��3����������Ϊǿ�����Ϊǿ�ᣬ�������1molH2O��1���ġ�H=-a KJ?mol-1��������
��4�����������������Ʒ�Ӧ�������������������������Ƶ����Ķ����йأ�
��1��C��Һ��μ��������������ʵ�����Ũ�ȵ�A��Һ���У�HCl��̼���Ʒ�Ӧ����NaHCO3������ˮ�����Ƚ�����Ũ�ȵĴ�С��
��2������������������������Ӧ��������ȫ����Ӧ�������Ӻ������֮�䰴�����ʵ���֮����2��3���з�Ӧ��
��3����������Ϊǿ�����Ϊǿ�ᣬ�������1molH2O��1���ġ�H=-a KJ?mol-1��������
��4�����������������Ʒ�Ӧ�������������������������Ƶ����Ķ����йأ�
����⣺��A��B��Һ�ʼ��ԣ��������ĸ��������У�̼���ˮ���Լ��ԣ��������м��ԣ�����A��B�к��е�������Ϊ̼�����������������Ϊ�����Ӻ�������ˮ�������ԣ������������ԣ�����C��D��E��Һ���е�������ΪH+��Al3+��Ag+��Na2CO3�Ϳ����Ե����η�Ӧ��ʵ���ǣ������Ӻ�̼�������֮��˫ˮ�ⷴӦ��������г����������壬����A��̼���ƣ�B������������̼���ƺ��ᷴӦ�����������̼���壬�������е������Ӻ���������ɵ������У��������ܺ����ᡢ�Ȼ��ơ�������������������Ӧ���ɳ���������D����������C�����ᣬEΪ��������
��1��C��Һ��μ��������������ʵ�����Ũ�ȵ�A��Һ���У�HCl��̼���Ʒ�Ӧ���ɵ����ʵ�����NaHCO3��NaCl��̼��������ӵ�ˮ����ڵ��룬��Һ�Լ��ԣ�
������Ũ��Ϊc��Na+����c��C1-����c��HCO3-����c��OH-����c��H+����c��CO32-����
�ʴ�Ϊ��c��Na+����c��C1-����c��HCO3-����c��OH-����c��H+����c��CO32-����
��2������������������������Ӧ�������Ӻ������֮�䰴�����ʵ���֮����2��3��
��2Al3++3SO42-+3Ba2++8OH-�T2AlO2-+4H2O+3BaSO4����
�ʴ�Ϊ��2Al3++3SO42-+3Ba2++8OH-�T2AlO2-+4H2O+3BaSO4����
��3����NaOH��aq��+HNO3��aq��=NaNO3��aq��+H2O��1������H=-a KJ?mol-1����������Ϊǿ�����Ϊǿ�ᣬ��B��C��ϡ��Һ��Ӧ���Ȼ�ѧ����ʽΪ
OH-��aq��+H+��aq��=H2O��1����H=-a KJ/mol��
Ba��OH��2��aq��+2HC1��aq��=
BaC12��aq��+H2O��1����H=-a KJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+H2O��1����H=-2a KJ/mol��
�ʴ�Ϊ��OH-��aq��+H+��aq��=H2O��1����H=-a KJ/mol��
Ba��OH��2��aq��+2HC1��aq��=
BaC12��aq��+H2O��1����H=-a KJ/mol
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+H2O��1����H=-2a KJ/mol��
��4�����������������Ƶķ�Ӧʵ��Ϊ��Al3++3OH-=Al��OH��3����Al��OH��3+OH-=[Al��OH��4]-��100mL 0.1mol/L����������Һ�У����������ӵ����ʵ���Ϊ0.02mol��
35mL 2mol/L NaOH��Һ�к��������������ʵ���Ϊ0.07mol��0.02mol����������0.06mol��������������0.02mol������������Ȼ��0.01mol�����������ֽ�0.01mol�����������ܽ⣬
����ʣ���������������ʵ�����0.01mol���ʴ�Ϊ��0.01mol��
��1��C��Һ��μ��������������ʵ�����Ũ�ȵ�A��Һ���У�HCl��̼���Ʒ�Ӧ���ɵ����ʵ�����NaHCO3��NaCl��̼��������ӵ�ˮ����ڵ��룬��Һ�Լ��ԣ�
������Ũ��Ϊc��Na+����c��C1-����c��HCO3-����c��OH-����c��H+����c��CO32-����
�ʴ�Ϊ��c��Na+����c��C1-����c��HCO3-����c��OH-����c��H+����c��CO32-����
��2������������������������Ӧ�������Ӻ������֮�䰴�����ʵ���֮����2��3��
��2Al3++3SO42-+3Ba2++8OH-�T2AlO2-+4H2O+3BaSO4����
�ʴ�Ϊ��2Al3++3SO42-+3Ba2++8OH-�T2AlO2-+4H2O+3BaSO4����
��3����NaOH��aq��+HNO3��aq��=NaNO3��aq��+H2O��1������H=-a KJ?mol-1����������Ϊǿ�����Ϊǿ�ᣬ��B��C��ϡ��Һ��Ӧ���Ȼ�ѧ����ʽΪ
OH-��aq��+H+��aq��=H2O��1����H=-a KJ/mol��
| 1 |
| 2 |
| 1 |
| 2 |
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+H2O��1����H=-2a KJ/mol��
�ʴ�Ϊ��OH-��aq��+H+��aq��=H2O��1����H=-a KJ/mol��
| 1 |
| 2 |
| 1 |
| 2 |
��Ba��OH��2��aq��+2HC1��aq��=BaC12��aq��+H2O��1����H=-2a KJ/mol��
��4�����������������Ƶķ�Ӧʵ��Ϊ��Al3++3OH-=Al��OH��3����Al��OH��3+OH-=[Al��OH��4]-��100mL 0.1mol/L����������Һ�У����������ӵ����ʵ���Ϊ0.02mol��
35mL 2mol/L NaOH��Һ�к��������������ʵ���Ϊ0.07mol��0.02mol����������0.06mol��������������0.02mol������������Ȼ��0.01mol�����������ֽ�0.01mol�����������ܽ⣬
����ʣ���������������ʵ�����0.01mol���ʴ�Ϊ��0.01mol��
���������⿼��֪ʶ��϶࣬�������ӵĹ��桢���ӵ��������ƶ������ǽ����Ĺؼ����ۺ���ǿ���ѶȽϴ�ѧ������Ϥˮ�⡢�Ȼ�ѧ��Ӧ����ʽ�����ӷ�Ӧ��֪ʶ�����

��ϰ��ϵ�д�
�����Ŀ
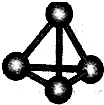 ����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��
����A��B��C��D��E��F���ֶ�����Ԫ�أ����ǵ�ԭ��������������D��E���⻯����ӹ��Ͷ���V�ͣ�A��B������������֮����C��������������ȣ�A�ֱܷ���B��C��D�γɵ���������ȵķ��ӣ���A��D���γɵĻ���������¾�ΪҺ̬��