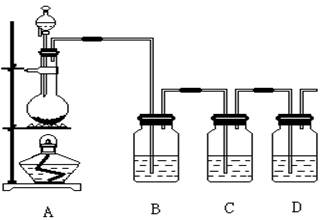
��Ŀ����
(����ʽÿ��2��,����ÿ��1��,��21�֡�)������ԭ��Ӧ�����ӷ�Ӧ�Ǹ�һ��ѧ������Ҫ��ѧ��Ӧ����,��ϸ˼���ش���������:
��������һ����ˮ����ķ�Ӧ:
��2Na + 2H2O =" 2NaOH" + H2��
��2Na2O2 + 2H2O =" 4NaOH" + O2��
��Na2O+ H2O ="2NaOH"
��3Fe+4H2O Fe3O4+4H2
Fe3O4+4H2
��2H2O 2H2��+ O2��
2H2��+ O2��
��2F2 + 2H2O =" 4HF" + O2
��1�����в�����������ԭ��Ӧ���� �����ţ�
��2������Щ������ԭ��Ӧ��,ˮֻ���������ķ�Ӧ�� �����ţ�
ˮֻ����ԭ���ķ�Ӧ�� �����ţ�ˮ����������,������ԭ���� �����ţ�
ˮ�Ȳ���������,�ֲ�����ԭ���� �����ţ�
��3�������������,������ԭ��Ӧ��ʵ���ǣ� ��
| A�������е�ԭ��������� | B����Ԫ�صĵ�ʧ |
| C�����ӵĵ�ʧ���õ��ӶԵ�ƫ�� | D�����ϼ۵ĸı� |
����д�����з�Ӧ�����ӷ���ʽ:
�����Ȼ�����Һ��������:
�ڹ���������̼ͨ�����ʯ��ˮ:
�����Ȼ�����Һ�м���������NaOH��Һ:
����д�����з�Ӧ�Ļ�ѧ����ʽ:
�ٶ�����������������Ӧ:
������������������Һ��Ӧ:
��ͭƬ��ϡ����ķ�Ӧ:
����1���� (1��) ��2���٢�;��;��;�� ��ѡ��1����1�֣���4�֣�
��3��C (1��) ��4���ǣ� NO2�� NO2����1�֣���3�֣�
����2Fe3+ + Fe = 3Fe2+ ;CO2 + OH- = HCO3- ; Al3+ + 4OH- = AlO2- + 2H2O
����2SO2 + O2 ��
�� 2SO3 ;Cl2 + 2NaOH =" NaCl" + NaClO + H2O;
2SO3 ;Cl2 + 2NaOH =" NaCl" + NaClO + H2O;
3Cu + 8HNO3(ϡ) = 3Cu(NO3)2 + 2NO��+ 4H2O
�����������������1�����в�����������ԭ��Ӧ�ļ����ϼ�û�з����仯�ķ�Ӧ��ֻ�Тۣ�
��2��ˮֻ���������ķ�Ӧ��ˮ�е�H���ϼ۽��ͣ�����Ӧ����H2��ѡ�٢ܣ�
ˮֻ����ԭ���ķ�Ӧ��ˮ�е�O���ϼ����ߣ�������Ӧ����O2��ͬʱO2������Դ��ˮ��ֻ��ѡ�ޣ�ˮ����������,������ԭ��������м�������������������ѡ�ݣ���������ԭ��Ӧ������ˮ�е����������ϼ�û�б仯����ֻ���Ǣڣ�
��3�������������,������ԭ��Ӧ��ʵ���ǵ��ӵĵ�ʧ���õ��ӶԵ�ƫ�ƣ����ֳ������ǻ��ϼ۵ĸı䣻
��4��3NO2+H2O �� 2HNO3+NO��N�Ļ��ϼ۷����˱仯����������ԭ��Ӧ��NO2��N��2��N���ϼ����ߵ�+5��HNO3����1��N���͵�+2�۵�NO��������������ԭ������NO2��
����2Fe3+ + Fe = 3Fe2+ ;CO2 + OH- = HCO3- ; Al3+ + 4OH- = AlO2- + 2H2O
����2SO2 + O2 ��
�� 2SO3 ;Cl2 + 2NaOH =" NaCl" + NaClO + H2O;
2SO3 ;Cl2 + 2NaOH =" NaCl" + NaClO + H2O;
3Cu + 8HNO3(ϡ) = 3Cu(NO3)2 + 2NO��+ 4H2O
���㣺����������ԭ��Ӧ�и���ķ���������������ԭ��Ӧ����ʽ����д��
������ѧ�����ڷ���ʽ����дһֱ���ѵ㣬��ƽʱ��дʱҪ����˼�����ϼ۵ı仯����ס�������ʵ������Ի�ԭ�ԣ��������ڷ���ʽ����д�����°빦�������á�

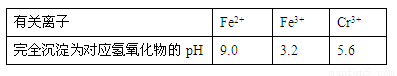
ij����С����MnO2��Ũ�����Ʊ�Cl2ʱ�����ø����չ�����SO2��NaOH��Һ����β���������մ�����
��1�������SO2��������NaOH��Һ��Ӧ�Ļ�ѧ����ʽ��
��2����ӦCl2+Na2SO3+2NaOH===2NaCl+Na2SO4+H2O��ÿת��2.5mol�ĵ�����μӷ�Ӧ�Ļ�ԭ�������ʵ���Ϊ mol��
��3������β��һ��ʱ�������Һ��ǿ���ԣ��п϶�����Cl![]() ��OH
��OH![]() ��SO
��SO![]() �������ʵ�飬̽��������Һ�п��ܴ��ڵ����������ӣ������ǿ�����CO2��Ӱ�죩��
�������ʵ�飬̽��������Һ�п��ܴ��ڵ����������ӣ������ǿ�����CO2��Ӱ�죩��
������������� ��
����1��ֻ����SO32-�� ����2��ֻ����ClO![]()
����3���Ȳ�����SO32-Ҳ������ClO![]() ��
��
����4�� ��
�����ʵ�鷽������ʵ�顣���ڴ����д��ʵ�鲽���Լ�Ԥ������ͽ���
��ѡʵ���Լ���3moL![]() L-1H2SO4��0.01mol
L-1H2SO4��0.01mol![]() L-1KMnO4����ɫʯ����Һ����ÿ��2�֣�
L-1KMnO4����ɫʯ����Һ����ÿ��2�֣�
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ��������Һ���Թ��У��μ�3 moL | |
| ����2�� | |
| ����3�� |
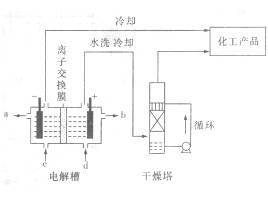
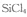 Ҳ��ת��Ϊ
Ҳ��ת��Ϊ ��ѭ��ʹ�á�һ�������£���200C�����ܱ������з�����Ӧ:
��ѭ��ʹ�á�һ�������£���200C�����ܱ������з�����Ӧ: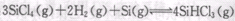 ����ƽ���H2��SiHCL3���ʵ���Ũ�ȷֱ�Ϊ0. 140mol/L��0. 020mol/L����H2ȫ����Դ�����ӽ���Ĥ���ĵ���������������Ĵ�NaCl�����ʵ���Ϊ_______ (2��)mol��
����ƽ���H2��SiHCL3���ʵ���Ũ�ȷֱ�Ϊ0. 140mol/L��0. 020mol/L����H2ȫ����Դ�����ӽ���Ĥ���ĵ���������������Ĵ�NaCl�����ʵ���Ϊ_______ (2��)mol��