��Ŀ����
��16�֣�����Ǧ��PbSO4���㷺Ӧ��������Ǧ���ء���ɫ���ϵȡ����÷�Ǧ��PbS��ֱ���Ʊ�����Ǧ��ĩ���������£�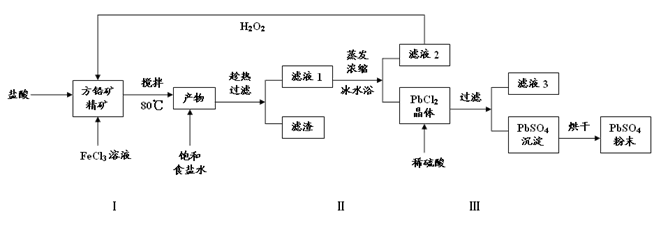
��֪��������PbCl2��s��+2Cl��(aq) PbCl4-(aq) ��H��0
PbCl4-(aq) ��H��0
������Ksp(PbSO4)=1.08��10-8, Ksp(PbCl2)=1.6��10-5
������Fe3����Pb2��������������ʽ��ȫ����ʱ����Һ��PHֵ�ֱ�Ϊ3.2��7.04
��1�������������PbCl2��S�����ӷ���ʽ �������������һ��Ŀ����Ϊ�˿���PHֵ��0.5��1.0��ԭ���� ��
��2���û�ѧƽ���ƶ���ԭ�����Ͳ������ʹ�ñ�ˮԡ��ԭ�� ��
��3��д��PbCl2����ת��ΪPbSO4���������ӷ���ʽ ��
��4���������ӷ���ʽ������Һ2����H2O2��ѭ�����õ�ԭ�� ����Һ3�� ��
��5��Ǧ���صĵ��Һ�����ᣬ���������缫�ϳ����� PbSO4�ֱ�ת��ΪPbO2��Pb�����ʱ�����ĵ缫��ӦʽΪ ��
��16�֣�
��1��PbS+2Fe3��+2Cl��= PbCl2+2Fe2��+S��3�֣�������Fe3����Pb2����ˮ�⣨2 �֣�
��2���ñ�ˮԡʹ��ӦPbCl2��s��+2Cl��(aq) PbCl4��(aq)�����ƶ���ʹPbCl4 ������ת��ΪPbCl2�������������3�֣�
PbCl4��(aq)�����ƶ���ʹPbCl4 ������ת��ΪPbCl2�������������3�֣�
��3��PbCl2��s��+SO42-(aq) PbSO4 (s) +2Cl��(aq) ��2�֣�
PbSO4 (s) +2Cl��(aq) ��2�֣�
��4��2Fe2��+ H2O2+2H��=2Fe3��+2H2O��2�֣������ᣨ2�֣�
��5��PbSO4+2e��="Pb+" SO42����2�֣�
���������������1�������������PbCl2��S��˵����������ᡢ�Ȼ�����PbS����������ԭ��Ӧ��SԪ�ػ��ϼ����ߣ�����Ԫ�صĻ��ϼ۽��ͣ����ӷ���ʽΪPbS+2Fe3��+2Cl��= PbCl2+2Fe2��+S��Fe3����Pb2��������������ʽ��ȫ����ʱ����Һ��PHֵ�ֱ�Ϊ3.2��7.04�����Լ����������һ��Ŀ����Ϊ�˿���PHֵ��0.5��1.0��ԭ��������Fe3����Pb2����ˮ�⣻
��2�����뱥��ʳ��ˮ�����PbCl2��s��+2Cl��(aq) PbCl4-(aq) ��H��0���ڱ�ˮԡ�У��¶Ƚϵͣ�ʹƽ�������ƶ�������PbCl2�����������
PbCl4-(aq) ��H��0���ڱ�ˮԡ�У��¶Ƚϵͣ�ʹƽ�������ƶ�������PbCl2�����������
��3��Ksp(PbSO4)=1.08��10-8< Ksp(PbCl2)=1.6��10-5�����ݳ�����ת����PbCl2����ת��ΪPbSO4��������ΪPbCl2�м���ϡ�����ƻ����Ȼ�Ǧ���ܽ�ƽ�⣬ʹ�ܽ�ƽ�������ƶ���������Ǧ�����ӷ���ʽΪPbCl2��s��+SO42-(aq) PbSO4 (s) +2Cl��(aq)��
PbSO4 (s) +2Cl��(aq)��
��4����Һ2����Ҫ�ɷ����Ȼ���������������ⷴӦ�������Ȼ�������������ʹ�ã����ӷ���ʽΪ2Fe2��+ H2O2+2H��=2Fe3��+2H2O���ɣ�3����֪�Ȼ�Ǧת��Ϊ����Ǧͬʱ�����Ȼ��⣬������Һ3��������Һ��
��5�����ʱ�ǰѵ���ת��Ϊ��ѧ�ܣ��൱�ڵ���װ�ã���������������ԭ��Ӧ��Pb�Ļ��ϼ۽��ͳ�Ϊ����Pb���缫��ӦʽΪPbSO4+2e��="Pb+" SO42��
���㣺��������̵ķ�������ѧƽ���Ӧ�ã��绯ѧԭ����Ӧ��

 �߽�������ϵ�д�
�߽�������ϵ�д�����Һ���ܴ��������һ�����ӻ������
| A��K+ ��Al3+��SO42����NH3��H2O | B��NH4+��H+��NO3����HCO3�� |
C��Na+ ��CH3COO����CO32����OH�� | D��Na+��K+��SO32����Cl2 |
������ط�Ӧ�����ӷ���ʽ��д��ȷ����
| A������������������Fe(OH)3+3H+ = Fe3++3H2O |
| B������ͭ��Һ�����ԣ�Cu2+ + 2H2O = Cu(OH)2��+ 2H+ |
C����̼�������Һ�мӹ���ʯ��ˮ�����ȣ�NH4++OH-  NH3��+H2O NH3��+H2O |
| D�����ữ�ĸ��������Һ����˫��ˮ��2MnO4-+6H++5H2O2 = 2Mn2++5O2��+8H2O |
����ѧ����ѡ��2����ѧ�뼼������15�֣�
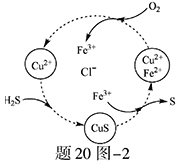
��Ƴ���ͭ��ˮ�к���CN-��Cr2O72-���ӣ���Ҫ������������ŷš��ó��ⶨ�������̽��з�ˮ�������ش��������⣺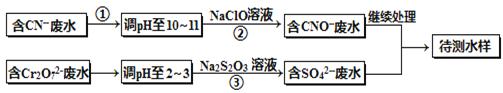
��1������������ˮ��������Ҫʹ�õķ�����_________________��
��2�����з�Ӧ��������ų����÷�Ӧ�����ӷ���ʽΪ______________��
��3��������У�ÿ����0.4mol Cr2O72-ʱת�Ƶ���2.4mol���÷�Ӧ�����ӷ���ʽΪ ��
��4��ȡ��������ˮ�����Թ��У�����NaOH��Һ���۲쵽����ɫ�������ɣ��ټ�Na2S��Һ����ɫ����ת���ɺ�ɫ��������ʹ�û�ѧ��������ֽ��Ͳ����������ԭ�� ��
��5��Ŀǰ��������Cr2O72-��ˮ������������巨���÷������ˮ�м���FeSO4 ��7H2O��Cr2O72-��ԭ��Cr3+������pH��Fe��Crת�����൱�ڣ� ����������,�������ֱ�ʾԪ�ؼ�̬���ij�����
����������,�������ֱ�ʾԪ�ؼ�̬���ij�����
����1mol Cr2O72-�������a mol FeSO4 ? 7H2O�����н�����ȷ����_______��
| A��x ="0.5" ,a ="8" | B��x ="0.5" ,a =" 10" | C��x =" 1.5" ,a =8 | D��x =" 1.5" ,a = 10 |
��������(ClO2)Ϊһ�ֻ���ɫ���壬�ǹ����Ϲ��ϵĸ�Ч�����ס����١���ȫ��ɱ����������
(1)��ҵ���Ʊ�ClO2�ķ�Ӧԭ��Ϊ2NaClO3��4HCl=2ClO2����Cl2����2H2O��2NaCl��
��Ũ�����ڷ�Ӧ����ʾ������������________��
| A��ֻ�л�ԭ�� | B����ԭ�Ժ����� |
| C��ֻ�������� | D�������Ժ����� |
(2)Ŀǰ�ѿ������õ�ⷨ��ȡClO2���¹��ա�
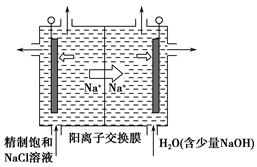
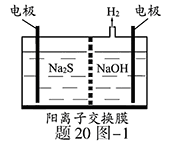
����ͼ����ʯī���缫��һ�������µ�ⱥ��ʳ��ˮ��ȡClO2��ʾ��ͼ������������ClO2�ĵ缫��ӦʽΪ_____________________________________��
�ڵ��һ��ʱ�䣬�������������������Ϊ112 mL(��״��)ʱ��ֹͣ��⡣ͨ�������ӽ���Ĥ�������ӵ����ʵ���Ϊ________mol����ƽ���ƶ�ԭ������������pH�����ԭ��______________________________________��
(3)ClO2����ˮ��Fe2����Mn2����S2����CN���������Ե�ȥ��Ч����ij������ˮ�к�CN��a mg/L������ClO2��CN���������������������������ɣ������ӷ�Ӧ����ʽΪ________________������100 m3������ˮ��������ҪClO2________mol��

 ��Һ����Al3+ǡ��ȫ������ʱ��
��Һ����Al3+ǡ��ȫ������ʱ��  S ��n��1��S+ S2��
S ��n��1��S+ S2��