��Ŀ����
����Ŀ���绯ѧԭ���ڷ�ֹ������ʴ������ת�������ʺϳɵȷ���Ӧ�ù㷺��
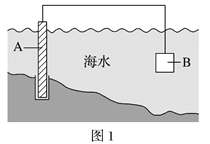
��1��ͼ1�У�Ϊ�˼�����ˮ�Ը�բ��A�ĸ�ʴ������B����ѡ��__________������ĸ��ţ�
a��̼�� b��п�� c��ͭ��
�õ绯ѧԭ�����Ͳ���B�趨�ڲ�ԭ��_______________��
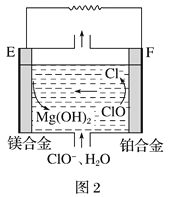
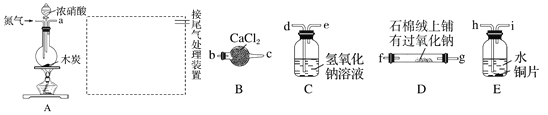
��2��þȼ�ϵ���ڿ��ƶ������豸��Դ�ͱ��õ�Դ�ȷ���Ӧ��ǰ��������ͼ2Ϊ��þ������������ȼ�ϵ��ԭ��ʾ��ͼ���缫Ϊþ�Ͻ�Ͳ��Ͻ�
��EΪ��ȼ�ϵ�ص�_____________������������������������F�缫�ϵĵ缫��ӦʽΪ_____________��
��þȼ�ϵ�ظ����������Ը�ʴ����������ʹ���������ʽ��ͣ��û�ѧ���������ԭ��____________��
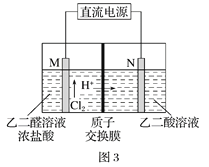
��3����ȩ�ᣨHOOC-CHO�����л��ϳɵ���Ҫ�м��塣��ҵ������˫���ҳɶԵ�ⷨ��������ȩ�ᣬԭ����ͼ3��ʾ����װ��������������Ϊ���Ե缫�������Ҿ��ɲ�����ȩ�ᣬ�����Ҷ�ȩ��M�缫�IJ��ﷴӦ������ȩ�ᡣ
��N�缫�ϵĵ缫��ӦʽΪ______________��
������2molH+ͨ�����ӽ���Ĥ������ȫ�����˷�Ӧ�����װ�������ɵ���ȩ��Ϊ________________mol��
���𰸡���1��b��п����ԭ��صĸ�������Zn -2e- = Zn2+�����������ܸ�ʴ���趨�ڲ�
��2���� ����ClO��+2e��+H2O = Cl��+2OH����
�� Mg+2H2O= Mg(OH)2+H2����
��3���� HOOC-COOH+2e��+2H+ ="HOOC-CHO+" H2O���� 2��
�������������������1���γ�ԭ���ʱ��Fe����������������Ҫѡ������Ա�Feǿ�Ľ���������������ѡп��п�Ļ����Ա�Feǿ�����������������ܸ�ʴ���趨�ڲ��ʴ�Ϊ��b��п����ԭ��صĸ�������ʧ���ӣ�Zn-2e-�TZn2+�����������ܸ�ʴ���趨�ڲ�
��2���١�þ-����������ȼ�ϵ����ʧ���ӵ�Ϊ��������MgΪ������������ClO-�õ������������ӣ��������ĵ缫��ӦʽΪ��ClO-+2e-+H2O�TCl-+2OH-���ʴ�Ϊ������ClO-+2e-+H2O�TCl-+2OH-��
��Mg�Ļ����Խ�ǿ����ˮ��Ӧ�����������䷴ӦΪ��Mg+2H2O�TMg��OH��2+H2�����ʴ�Ϊ��Mg+2H2O�TMg��OH��2+H2����
��3����N�缫��HOOC-COOH�õ�������HOOC-CHO����缫��ӦʽΪHOOC-COOH+2e-+2H+ �THOOC-CHO+H2O���ʴ�Ϊ��HOOC-COOH+2e-+2H+�THOOC-CHO+H2O��
��2mol H+ͨ�����ӽ���Ĥ��������ת��2mol���ӣ����ݵ缫����ʽHOOC-COOH+2e-+2H+ �THOOC-CHO+H2O����֪����1mol��ȩ�ᣬ��������������ȩ�������������ɵ���ȩ��Ϊ2mol���ʴ�Ϊ��2��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�