��Ŀ����
ij�¶��£���һ�̶��ݻ����ܱ�������Ͷ��һ�����ʵ�����N2��H2���п��淴Ӧ��N2(g)��3H2(g)  2NH3(g) ��2 minʱ�ﵽƽ��״̬����ʱc(N2)��5.00 mol/L��c(H2)��10.00mol/L��c(NH3)��5.00 mol/L������д��������̣�
2NH3(g) ��2 minʱ�ﵽƽ��״̬����ʱc(N2)��5.00 mol/L��c(H2)��10.00mol/L��c(NH3)��5.00 mol/L������д��������̣�
��1�����¶��£���Ӧ��ƽ�ⳣ���Ƕ��٣�
��2��H2����ʼŨ�ȣ�
��3����N2��ʾ�÷�Ӧ�ķ�Ӧ�����Ƕ��٣�
��4��N2��ת���ʣ�������λ��Ч���֣���
 2NH3(g) ��2 minʱ�ﵽƽ��״̬����ʱc(N2)��5.00 mol/L��c(H2)��10.00mol/L��c(NH3)��5.00 mol/L������д��������̣�
2NH3(g) ��2 minʱ�ﵽƽ��״̬����ʱc(N2)��5.00 mol/L��c(H2)��10.00mol/L��c(NH3)��5.00 mol/L������д��������̣���1�����¶��£���Ӧ��ƽ�ⳣ���Ƕ��٣�
��2��H2����ʼŨ�ȣ�
��3����N2��ʾ�÷�Ӧ�ķ�Ӧ�����Ƕ��٣�
��4��N2��ת���ʣ�������λ��Ч���֣���
��1��K=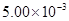
��2��
��3��
��4��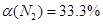
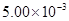
��2��

��3��

��4��
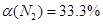
�����������1��
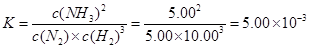
��2���ٶ�����Ϊ1L����ﵽƽ���
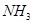 Ϊ5.00mol,���ݻ�ѧƽ���ϵʽ��֪������2.50mol��
Ϊ5.00mol,���ݻ�ѧƽ���ϵʽ��֪������2.50mol��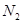 ��7.50mol��
��7.50mol�� ��7.50+10.00=17.50mol,����H2����ʼŨ��
��7.50+10.00=17.50mol,����H2����ʼŨ�� ��
����3���������

��4����ϣ�2��


��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
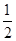 O2(g)
O2(g) SO3(g)����H����98 kJ/mol��ij�¶��£���һ���Ϊ2 L���ܱ������г���0.2 mol SO2��0.1 mol O2,5 min��ﵽƽ�⣬���ų�����11.76 kJ������˵����ȷ����(����)
SO3(g)����H����98 kJ/mol��ij�¶��£���һ���Ϊ2 L���ܱ������г���0.2 mol SO2��0.1 mol O2,5 min��ﵽƽ�⣬���ų�����11.76 kJ������˵����ȷ����(����) 2C(g)+2D(g)���ڲ�ͬ����²�÷�Ӧ���ʣ����з�Ӧ���������ǣ� ��
2C(g)+2D(g)���ڲ�ͬ����²�÷�Ӧ���ʣ����з�Ӧ���������ǣ� ��  2C(g)+2D(g) ��H��2minĩ�ﵽƽ�⣬����0.8mol D��
2C(g)+2D(g) ��H��2minĩ�ﵽƽ�⣬����0.8mol D�� =____________��
=____________��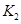 =l�����H________0���>����<������
=l�����H________0���>����<������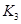 �� ��K3________
�� ��K3________ (A)=
(A)=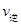 (C) D����������ܶȲ���
(C) D����������ܶȲ��� 4CO2(g)+N2(g)����H=��1200 kJ��mol��1
4CO2(g)+N2(g)����H=��1200 kJ��mol��1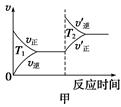
 CO2(g)+H2(g) ��H<0��820 ��ʱ�ڼס��ҡ������������ܱ������У���ʼʱ�����ұ�����Ͷ�ϣ��ﵽƽ��״̬ʱK=1.0��
CO2(g)+H2(g) ��H<0��820 ��ʱ�ڼס��ҡ������������ܱ������У���ʼʱ�����ұ�����Ͷ�ϣ��ﵽƽ��״̬ʱK=1.0��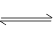 C(g)+D(g)��Ӧ��ƽ�ⳣ�����¶ȵĹ�ϵ���£�
C(g)+D(g)��Ӧ��ƽ�ⳣ�����¶ȵĹ�ϵ���£�