��Ŀ����
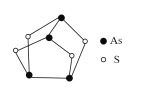
����Ŀ��ij���ӵĽṹ��ͼ��ʾ��-R Ϊ������������ A��B��D ����Ԫ��λ��Ԫ�����ڱ���ͬһ����������ڵ����ڣ�A �ķǽ����Դ��� B��D �� G �γɵ� DG3 �ڹ�ҵ�Ͽ�����Ư��ɱ��������A �� G �γɵ� AG3 ����ȫˮ�⣬��ˮ��IJ���֮һ H3AO3 ���������ϼ��ƽ����Ļ�ԭ����

(1)����δ�ɶԵ��ӵ�ԭ�ӻ���Ӿ��д��ԡ�D ��ij�������� D2O4 �Ĵ��Դ�С���¶ȳ�����ع�ϵ�����������¶ȵ����������� D2O4 ![]() 2DO2����H______0��������������������=������
2DO2����H______0��������������������=������
(2)DG3 ����ɱ�������� HGO ��ȣ�DG3 �ɴ���ӳ�ɱ��������ʱ�䣬�Դӷ�Ӧ�������ۺ�ƽ���ƶ�����������ѡ��һ����������ԭ��____________________________��
(3)���������еķ��ǻ��ⲻ�ܷ������롣H3AO3 ������ A ԭ�������ĵ��Ӷ������˹��ۼ����γɣ����÷���ʽ��ʾ H3AO3 ��������Һ�ʼ��Ե�ԭ��_____��
(4)Һ���������2NH3(1) ![]() NH4++NH2-�ɵ��磬Һ̬ D2O4 ��Ҳ���� D2O4
NH4++NH2-�ɵ��磬Һ̬ D2O4 ��Ҳ���� D2O4![]() DO++DO3-�������������̵ı�������Ϊ___________��
DO++DO3-�������������̵ı�������Ϊ___________��
(5)T��ʱ����һ���Ϊ VL ���ܱ������з���һ������ ACl5 ���壬����ʽ������Ӧ��ACl5��s��![]() ACl3��g��+Cl2��g������H>0����������������ѹǿ�仯���±���
ACl3��g��+Cl2��g������H>0����������������ѹǿ�仯���±���
ʱ�� t/s | 0 | 5 | 10 | 15 | 20 | 25 | 30 | �� |
��ѹ P/kPa | 0 | 1.0 | 2.0 | 3.0 | 4.0 | 5.0 | 5.0 | 5.0 |
���������£��Է�ѹ��ʾ��ƽ�ⳣ�� Kp=_____(kPa)2��������������λС������ �������¶Ȳ��䣬30s ʱ��������ѹ������ƽ��������ڵ���ѹ��_____�����������������������������������������������ɾ�����������ѹ�������ڵ���ѹ��_____������������������������������������
���𰸡�> NCl3��ˮ��Ӧ������̫С���� NCl3��ˮ��Ӧ��ƽ�ⳣ��̫С�� HPO32-+ H2O![]() H2PO3-+OH- ǰ��δ��������ת�ƣ����߷����˵���ת�� 6.25 ���� ����
H2PO3-+OH- ǰ��δ��������ת�ƣ����߷����˵���ת�� 6.25 ���� ����
��������
A��B��D ����Ԫ��λ��Ԫ�����ڱ���ͬһ����������ڵ����ڣ�A �ķǽ����Դ��� B��D �� G �γɵ� DG3 �ڹ�ҵ�Ͽ�����Ư��ɱ����������DΪN��GΪCl��AΪP��PCl3 ����ȫˮ�⣬��ˮ��IJ���֮һ H3PO3 ���������ϼ��ƽ����Ļ�ԭ������AΪP��BΪAs��DΪN,GΪCl��
��1��DΪNԪ�أ�D ��ij�������� N2O4 �Ĵ��Դ�С���¶ȳ�����ع�ϵ�����������¶ȵ���������NO2����δ�ɶԵ��ӣ�����δ�ɶԵ��ӵ�ԭ�ӻ���Ӿ��д��ԣ������ߣ�N2O4 ![]() 2NO2Ҫ������������ N2O4
2NO2Ҫ������������ N2O4 ![]() 2NO2����H > 0��
2NO2����H > 0��
�ʴ�Ϊ�� > ��
��2��NCl3 ����ɱ�������� HClO ��ȣ�NCl3 �ɴ���ӳ�ɱ��������ʱ�䣬�ӷ�Ӧ��������NCl3��ˮ��Ӧ������̫С����ƽ���ƶ����۽�����ԭ��NCl3��ˮ��Ӧ��ƽ�ⳣ��̫С��
�ʴ�Ϊ��NCl3��ˮ��Ӧ������̫С���� NCl3��ˮ��Ӧ��ƽ�ⳣ��̫С����
��3�����������еķ��ǻ��ⲻ�ܷ������롣H3PO3 ������ P ԭ�������ĵ��Ӷ������˹��ۼ����γɣ�H3PO3�ĽṹʽΪ��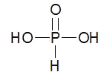 �Ƕ�Ԫ�ᣬ�����ᣬ����ˮ�⣬��Һ�ʼ��ԣ��÷���ʽ��ʾ H3AO3 ������HPO32-��Һ�ʼ��Ե�ԭ��HPO32-+ H2O
�Ƕ�Ԫ�ᣬ�����ᣬ����ˮ�⣬��Һ�ʼ��ԣ��÷���ʽ��ʾ H3AO3 ������HPO32-��Һ�ʼ��Ե�ԭ��HPO32-+ H2O![]() H2PO3-+OH-��
H2PO3-+OH-��
�ʴ�Ϊ��HPO32-+ H2O![]() H2PO3-+OH-��
H2PO3-+OH-��
��4��Һ���������2NH3(1) ![]() NH4++NH2-�ɵ��磬N�Ļ��ϼ۲��䣬Һ̬ N2O4 ��Ҳ���� N2O4
NH4++NH2-�ɵ��磬N�Ļ��ϼ۲��䣬Һ̬ N2O4 ��Ҳ���� N2O4![]() NO++NO3-��N2O4��NΪ+4�ۣ�NO+��NΪ+3�ۣ�NO3-��NΪ+5�ۣ������������̵ı�������Ϊ��ǰ��δ��������ת�ƣ����߷����˵���ת�ơ�
NO++NO3-��N2O4��NΪ+4�ۣ�NO+��NΪ+3�ۣ�NO3-��NΪ+5�ۣ������������̵ı�������Ϊ��ǰ��δ��������ת�ƣ����߷����˵���ת�ơ�
�ʴ�Ϊ��ǰ��δ��������ת�ƣ����߷����˵���ת�ƣ�
��5��T��ʱ����һ���Ϊ VL ���ܱ������з���һ������ PCl5 ���壬����ʽ������Ӧ��PCl5��s��![]() PCl3��g��+Cl2��g������H>0����������������ѹǿ�仯��ƽ��ʱ��ѹǿΪ5.0kPa��PCl3��g����Cl2��g���ķ�ѹ��Ϊ2.5kPa�����������£��Է�ѹ��ʾ��ƽ�ⳣ�� Kp=p(PCl3)p(Cl2)=
PCl3��g��+Cl2��g������H>0����������������ѹǿ�仯��ƽ��ʱ��ѹǿΪ5.0kPa��PCl3��g����Cl2��g���ķ�ѹ��Ϊ2.5kPa�����������£��Է�ѹ��ʾ��ƽ�ⳣ�� Kp=p(PCl3)p(Cl2)=![]() =6.25 (kPa)2��������������λС������
=6.25 (kPa)2��������������λС������
�������¶Ȳ��䣬ƽ�ⳣ�����䣬30s ʱ��������ѹ��ƽ�������ƶ�������ƽ��������ڵ���ѹ�����䣻
�����������ɾ�����������ѹ��ƽ�������ƶ���������ȣ�����ƽ����������¶����ߣ������ڵ���ѹ�����ߡ�
�ʴ�Ϊ��6.25������ �����ߡ�

 �Ķ��쳵ϵ�д�
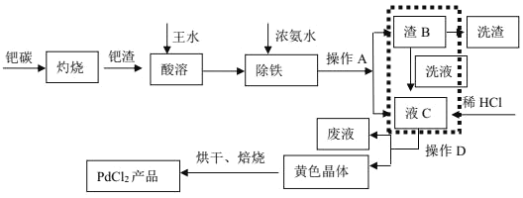
�Ķ��쳵ϵ�д�����Ŀ���й���ϡ����Դʮ�ַḻ���п��ɼ�ֵ�Ĵ����������һ��ijʵ��С�������ú������衢�������ʵķ�̼����(��Ҫ�ɷ�ΪCeFCO3)�Ʊ����Ȼ��棬�����ͼ��ʾ���̣�
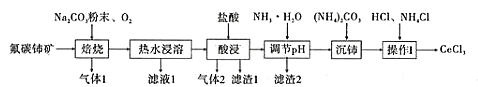
��֪����Ce2O3�ڼ����������ܱ���������������������ˮ������ǿ�����Ե�CeO2��
��M(CeCl3)=246.5g/mol����ˮ��Һ��Ce4+�ܱ���ԭ��Ce3+��
��0.1mol��L-1�����������γ��������������pH���£�
���������� | Fe3+ | Ce3+ | Al3+ |
��ʼ������pH | 1.5 | 7.6 | 3.4 |
������ȫ��pH | 2.8 | 9.4 | 4.7 |
�ش��������⣺
(1)��̼�������Ļ��ϼ�Ϊ______������1�Ļ�ѧʽΪ__________��
(2)���շ�̼�������CeO3�Ļ�ѧ����ʽ��____________��
(3)���ʱ��������2�����ӷ�Ӧ����ʽ��_______��
(4)Ϊ��������������2��ȥ����NH3��H2O�����������Һ��pH����ܳ���______��
(5)������õ���Ce2(CO3)3��nH2Oϴ�Ӹɾ���������в���I���������֤��Ce2(CO3)3��nH2O�Ѿ�ϴ�Ӹɾ�________��
(6)����I�ǽ��õ���Ce2(CO3)3��nH2O��������������ȫ�ܽ⣬�Ⱦ�������Ũ������ȴ�ᾧ�����ˡ�ϴ�ӡ�����õ�CeCl3��7H2O���ٽ��þ�����NH4Cl��������������ȣ��õ���ˮCeCl3�����У�NH4Cl��������___________��
(7)��Ʒ���Ȳⶨ��ȷ��ȡCeCl3��Ʒ0.3750g����ƿ�У�������ʵ���������Ce3+ǡ����ȫ����ΪCe4+������0.1000mol/L��(NH4)2Fe(SO4)2 ����Һ�ζ����յ㣬�ظ�3��ʵ�飬��Һ����������ֱ�Ϊ12.48mL��12.50mL��12.52mL�������Ʒ����������Ϊ_______(����1λС��)��
����Ŀ���п�Ժһ�����³ɹ�ʵ���˼����Ч������ϩ�������ڴ����������⣬�������о����ɻ�ż����Ӧ������ϩ���䷴Ӧ���£�2CH4(g)C2H4(g)��2H2(g)����H��0
��ѧ�� | H��H | C��H | C��C | C��C |
E(kJ/mol) | a | b | c | d |
��1����֪��ػ�ѧ���ļ������ϱ��������Ʊ���ϩ��Ӧ�Ħ�H��____________ (�ú�a��b��c��d�Ĵ���ʽ��ʾ)��
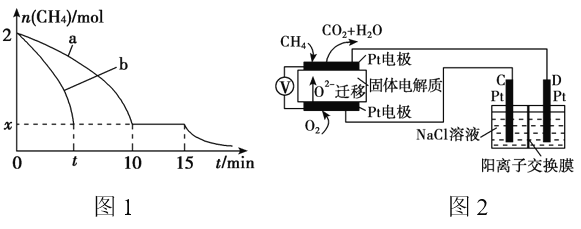
��2��T1�¶�ʱ����1 L�ĺ��ݷ�Ӧ���г���2mol CH4 ��������������Ӧ����Ӧ������ 0��15min CH4�����ʵ�����ʱ��仯��ͼ1�����10��15minʱH2��Ũ��Ϊ1.6mol/L��
��0��10min��CH4��ʾ�ķ�Ӧ����Ϊ__________mol/(L��min)��
����ͼ1������a������b�ֱ��ʾ���¶�T1ʱ��ʹ��������ͬ���������ͬ�Ĵ���ʱ���ﵽƽ�������n(CH4)�仯���ߣ����б�ʾ����������ϴ�������� ________ (�a���� ��b��)��
��15minʱ�����ı���練Ӧ����������n(CH4)����ͼ1����ʾ�仯����ı������������_____________________________________(�δ�һ������)��
��3��ʵ����v����k��c2(CH4)��v����k��c(C2H4)��c2(H2)����k����k��Ϊ���ʳ��������¶��йأ�T1�¶�ʱk����k���ı�ֵΪ______ (����ֵ)�������¶���T1���ߵ�T2����Ӧ��������ı���v��____v��(�>����������<��)���жϵ�������_________________ ��
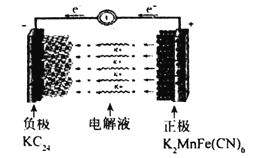
��4��������Ա����˼���ȼ�ϵ�ز����ڵ�⡣��ͼ2��ʾ��������Dz����� Y2O3�� ZrO2�Ĺ��壬���ڸ����´���O2����
��C����PtΪ______ ��(����������� )��
�ڸõ�ع���ʱ������Ӧ����ʽΪ_____________________��
���øõ�ص�ⱥ��ʳ��ˮ��һ��ʱ����ռ�����������������Ϊ112mL����������������Һ��25��ʱpH��_______ (������ǰ��NaCl��Һ�������Ϊ500mL)��