��Ŀ����
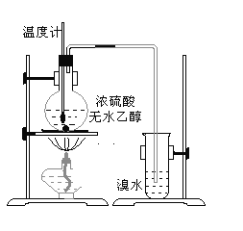
����Ŀ��Ϊ̽��ʵ��������ϩ����ϩ����ˮ�ļӳɷ�Ӧ����ͬѧ�������ͼ��ʾ��ʵ��װ�ã���������ʵ�顣���¶�����170������ʱ���д����������������������ͨ����ˮ�У���ˮ����ɫѸ����ȥ����ͬѧ��Ϊ�ﵽ��ʵ��Ŀ�ġ���ͬѧ��ϸ�۲��˼�ͬѧ������ʵ����̣����ֵ��¶�����100������ʱ����ɫҺ�忪ʼ��ɫ����160������ʱ�����Һȫ�ʺ�ɫ����170�泬�������������ٶ����Լӿ죬���ɵ������д̼�����ζ���ɴ����Ƴ���������������Ӧ�����ʣ�����Ӱ����ϩ�ļ���������ȥ���ݴ˻ش��������⣺
��1��д����ͬѧʵ������Ϊ�ﵽʵ��Ŀ�ĵ�������Ӧ�Ļ�ѧ����ʽ:__________________��
��2����ͬѧ�۲쵽�ĺ�ɫ������__________���̼���������__________����ͬѧ��Ϊ�̼�������Ĵ��ھͲ�����Ϊ��ˮ��ɫ����ϩ�ļӳɷ�Ӧ��ɵġ�ԭ���ǣ��û�ѧ���̱�ʾ����________________________________ ��
��3����ͬѧ���ݼ���ͬѧ�ķ�������Ϊ��������CO��CO2�������������Ϊ֤��CO���ڣ�����������¹��̣��ù��̿ɰ�ʵ���в������л����������������������徭��ȼ����ɫ���棬ȷ����һ����̼��
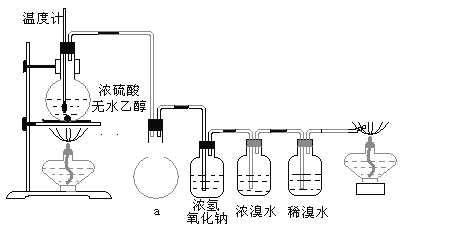
�����װ��a��������_____________________________________________
��Ũ��ˮ��������_________________________________________________��
ϡ��ˮ��������___________________________________________________��
���𰸡���11�֣��� CH3CH2OH![]() CH2��CH2����H2O ��2�֣�
CH2��CH2����H2O ��2�֣�
CH2��CH2��Br2��Br��CH2CH2��Br ��2�֣�
�� C��1�֣���SO2��1�֣���Br2��SO2��2H2O��2HBr��H2SO4��2�֣�
�Ǣ���ȫװ�ã���������1�֣�
��������ϩ�������������壨1�֣���������ϩ���������������Ƿ������1�֣�
��������
���⣨1�������Ҵ���Ũ����Ĵ������·�����������ˮ��ȡ��ϩ���Ҵ���������ȥ��Ӧ��CH3CH2OH![]() CH2==CH2��+ H2O����ˮ�е�������ϩ�����ӳɷ�Ӧ����CH2=CH2+Br2��CH2BrCH2Br��
CH2==CH2��+ H2O����ˮ�е�������ϩ�����ӳɷ�Ӧ����CH2=CH2+Br2��CH2BrCH2Br��
��2����ͬѧ��ϸ�۲��˼�ͬѧ������ʵ����̣����ֵ��¶�����100������ʱ����ɫҺ�忪ʼ��ɫ����160������ʱ�����Һȫ�ʺ�ɫ��Ũ�������ǿ�����ԣ��������Ҵ������Ҵ�������̼��ͬʱ��������ԭ�ɶ���������ˮ�����������������ԭ��Ӧ���屻����������+4�۵���ԭ��-1�۵��壬+4�۵�����������+6�۵�����ˮ��ɫ������ʽΪ��Br2+ SO2+ 2H2O ="=" 2HBr + H2SO4 ���ʴ�Ϊ��C��SO2��Br2+ SO2+ 2H2O ="=" 2HBr + H2SO4 ��
��3����ϩ����ˮ�е��巢���ӳɷ�Ӧ����ˮ��ɫ���ɼ�����ϩ�Ĵ��ڣ�����������巢��������ԭ��Ӧ��������������ᣬ�ɼ����������Ĵ��ڣ������װ��a�������ǣ���ȫװ�ã�����������Ũ��ˮ�������ǣ�������ϩ��������ϩ�Ͷ����������壩��ϡ��ˮ�������ǣ�������ϩ�Ƿ������

 ���Ǽ���С����ϵ�д�
���Ǽ���С����ϵ�д� �Ͻ�ƽ���Ȿϵ�д�
�Ͻ�ƽ���Ȿϵ�д�����Ŀ��ij��ҵ��ˮ�н����±������е�5��(������ˮ�ĵ��뼰���ӵ�ˮ��)���Ҹ������ӵ����ʵ���Ũ����ȣ���Ϊ0.1 mol/L��
������ | K����Mg2����Fe3����Al3����Fe2�� |
������ | Cl���� |
ijͬѧ��̽����ˮ����ɣ�����������ʵ�飺
���ò�˿պȡ������Һ���ڻ��������գ�����ɫ����(����ɫ�ܲ����۲�)��
��ȡ������Һ������KSCN��Һ�����Ա仯��
����ȡ��Һ�����������ᣬ����ɫ�������ɣ�����ɫ������������ɺ���ɫ����ʱ��Һ��Ȼ���壬����Һ������������䡣
�����������õ���Һ�м���BaCl2��Һ���а�ɫ�������ɡ�
���ƶϣ�
(1)�������������жϣ���Һ��һ�������е���������______________��һ�������е���������_____________(д���ӷ���)��
(2)���м�����������������ɫ��������ӷ���ʽ��___________________________���������ɰ�ɫ���������ӷ���ʽ��___________________________��
(3)���������ú���ɫ����ͨ��ˮ�У��������ɫ����������Ӧ�Ļ�ѧ����ʽΪ__________________________��
(4)��ͬѧ����ȷ��ԭ��Һ��������������___________����������________________д���ӷ���)��