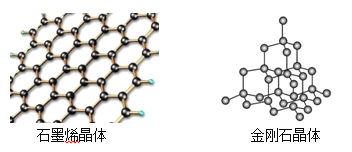
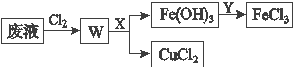
��Ŀ����
����Ŀ��ʯ���ǹ�ҵ��ѪҺ�������ǵ�����������ϢϢ��ء����������գ�
��1���������Ƶ����͵ķ�����___��
��2����ϩ����Ҫ��ʯ�ͻ�����Ʒ������ϩ��Ϊͬϵ�����___��(ѡ����)
A��CH3CH=CH2 B��CH2=CHCH=CH2
C��CH��CH D��CH3CH3
��3������ϩ��ȫ������������ʳƷ��װ��������ϩ�Ľṹ��ʽΪ___��
��4��������ϩ��;�㷺���䵥��Ľṹ��ʽΪ___����д������Ȳ��ȡ������ϩ�Ļ�ѧ����ʽ___����Ȳ���ɾۺϵõ���һ�ָ߷��ӻ�������������Ȳ������Ȳ___����(ѡ������������������)��
��5����Ȳ�ڲ�ͬ�Ĵ������¼���ɵõ���ͬ�IJ����д����Ȳ�ĵ���ʽ��___��
������������Ȳ����õ���ij�ֲ��ﻥΪͬϵ�����___��(ѡ����)��
A��C2H4 B��C3H8 C��![]() D��CH2=CHCH3
D��CH2=CHCH3
��6����Ȳ��ʹ��ˮ��ɫ��˵����Ȳ�ܷ���___��Ӧ���Ӧ���ͣ�����Ȳ��ʹ���Ը��������Һ��ɫ��˵����Ȳ���ױ�___��
��7����Ȳ��һ�����������۵õ�CH2=CH��C��C��CH=CH2��д����������������ͬ��ѧʽ�Ҳ������ӳɷ�Ӧ�����ʵĽṹ��ʽ___�������ʵ�ͬϵ���б�����һ�ȴ���ֻ��һ�ֵ����ʵĽṹ��ʽΪ___����һ��������
���𰸡�ʯ���ѻ� A ![]() CH2=CHCl CH��CH+HCl
CH2=CHCl CH��CH+HCl![]() CH2=CHCl ��
CH2=CHCl �� ![]() BD �ӳ� ����
BD �ӳ� ���� ![]()
![]() ��
��
��������
(1) ʯ�������У������ѻ���������͵IJ�����
(2)�ṹ���ƣ������������ɸ�CH2ԭ���ŵ��л��ﻥΪͬϵ�
(3)��ϩ�Ӿ۷�Ӧ���ɾ���ϩ��
(4) ������ϩ����Ľṹ��ʽΪCH2=CHCl������Ȳ��ȡ����ϩ���ڼӳɷ�Ӧ��
(5)��Ȳ�ĵ���ʽΪ![]() ��
��
�ṹ���ƣ������������ɸ�CH2ԭ���ŵ��л��ﻥΪͬϵ�
��6����Ȳ��ʹ��ˮ��ɫ����ʹ���Ը��������ɫ��
��7�����ݲ����Ͷȼ��ṹ�����жϡ�
(1) ʯ�������У������ѻ���������͵IJ����������ѽ��Ʊ���̬������������ϩ�ȣ����������Ƶ����͵ķ�����ʯ���ѻ���
(2) A. CH3CH=CH2������ϩ�ṹ���ƣ���������һ��CH2ԭ���ţ�����ϩ��Ϊͬϵ�ѡ��A��ȷ��
B. CH2=CHCH=CH2�����ڷ����к���2��̼̼˫��������ϩ�ṹ�����ƣ�����ϲ������CH2ԭ���ţ�����ϩ����ͬϵ�ѡ��B����
C. CH��CH�����ڷ����к���̼̼����������ϩ�ṹ�����ƣ�����ϲ������CH2ԭ���ţ�����ϩ����ͬϵ�ѡ��C����
D. CH3CH3��������û��̼̼˫��������ϩ�ṹ�����ƣ�����ϲ������CH2ԭ���ţ�����ϩ����ͬϵ�ѡ��D����
��ѡA��
(3)��ϩ�Ӿ۷�Ӧ���ɾ���ϩ������ϩ�Ľṹ��ʽΪ![]() ��
��
(4) ������ϩ����Ľṹ��ʽΪCH2=CHCl������Ȳ��ȡ����ϩ���ڼӳɷ�Ӧ����ѧ��Ӧ����ʽΪCH��CH+HCl![]() CH2=CHCl������Ȳ��̼̼������̼̼˫��������֣���һ�ֵ���ۺ���ܵ��磻
CH2=CHCl������Ȳ��̼̼������̼̼˫��������֣���һ�ֵ���ۺ���ܵ��磻
(5)��Ȳ�ĵ���ʽΪ![]() ��
��
��Ȳ�ڲ�ͬ�Ĵ������¼���ɵõ���ϩ�����飬����A.C2H4Ϊ��ϩ��ѡ��A����
B.C2H6Ϊ���飬C3H8������ṹ���ƣ��ڷ�����������1��CH2������ͬϵ�ѡ��B��ȷ��
C.![]() ����ϩ������ṹ�������ƣ�������ͬϵ�ѡ��C����
����ϩ������ṹ�������ƣ�������ͬϵ�ѡ��C����
D��CH2=CHCH3����ϩ�ṹ���ƣ��ڷ�����������1��CH2������ͬϵ�ѡ��D��ȷ��
��ѡBD��
��6����Ȳ��ʹ��ˮ��ɫ������Ϊ��ϩ�к���̼̼˫�����ܹ�����ˮ�����ӳɷ�Ӧ����Ȳ��ʹ���Ը��������ɫ������Ϊ��ϩ�к���̼̼˫�������������������
�ʴ�Ϊ���ӳɣ�������
��7��CH2=CH-C��C-CH=CH2�ķ���ʽΪC6H6�������Ͷ�Ϊ4���������ӳɷ�Ӧ��˵�������е��͵�˫�������������Խṹ��ʽΪ �������ʵ�ͬϵ���б�����һ�ȴ���ֻ��һ�ֵ����ʵ�Ϊ���ȡ���ڱ������γɸ߶ȶԳƵı���ͬϵ���Զ��ױ���1��3��5-���ױ��ȣ��ṹ��ʽ�ֱ�Ϊ
�������ʵ�ͬϵ���б�����һ�ȴ���ֻ��һ�ֵ����ʵ�Ϊ���ȡ���ڱ������γɸ߶ȶԳƵı���ͬϵ���Զ��ױ���1��3��5-���ױ��ȣ��ṹ��ʽ�ֱ�Ϊ![]() ��
�� ��
��

����Ŀ������������������������ж��ַ�����
��1���û���̿��ԭ����ij�о�С����ij�ܱ������м��������Ļ���̿��NO��������Ӧ��C(s)��2NO(g) ![]() N2(g)��CO2(g) ��H��Q kJ��mol��1 �� T1���£���Ӧ���е���ͬʱ���ø����ʵ�Ũ�Ȳ����������£�
N2(g)��CO2(g) ��H��Q kJ��mol��1 �� T1���£���Ӧ���е���ͬʱ���ø����ʵ�Ũ�Ȳ����������£�
ʱ�䣨min�� Ũ��mol/L | 0 | 10 | 20 | 30 | 40 | 50 |
NO | 1.00 | 0.40 | 0.20 | 0.30 | 0.30 | |
N2 | 0 | 0.40 | 0.60 | 0.60 | ||
CO2 | 0 | 0.60 | 0.60 |
��0��10 min �ڣ�N2��ƽ����Ӧ���� ��(N2)=________��T1��ʱ���÷�Ӧ��ƽ�ⳣ�� K=_____��
��30 min ����ֻ�ı�һ����������Ӧ���´ﵽƽ��ʱ�����ʵ�Ũ�����ϱ���ʾ����ı������������____________ ������ĸ��
a. ����һ�����Ļ���̿ b. �ı䷴Ӧ��ϵ���¶� c. ��С��������� d. ͨ��һ������NO
����30 min�������¶��� T2�棬���´ﵽƽ��ʱ��������NO��N2��CO2��Ũ��֮��Ϊ7��3��3����÷�Ӧ�ġ�H______0���������� �� �� ���� ���������� ��
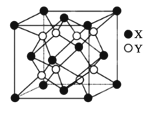
��2��NH3����ԭ����ԭ����ͼ��ʾ

���������� c(NO2)�� c(NO)=1��1��������ͼ����ʾ���ѵ���Ӧʱ����Ӧ������ת��1.5mol����ʱ�ų�������Ϊ 113.8 kJ���������ѵ���Ӧ���Ȼ�ѧ����ʽΪ_____ ��
��ͼ������һ��ʱ���ڣ�ʹ�ò�ͬ����Mn��Cr�ڲ�ͬ�¶��¶�Ӧ���ѵ��ʣ���ͼ��֪��ҵʹ�õ���Ѵ�������Ӧ�¶ȷֱ�Ϊ___��ʹ��Mn������ʱ���ѵ���b��a�γ�����ͼ�仯�Ŀ���ԭ����___��
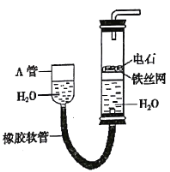
��3��ֱ�ӵ������Ҳ��������һ�ַ�������6����ϡ��������NOx���������ᣬ�ٽ�����Һ������۵�⣬ʹ֮ת��Ϊ���ᡣ���װ����ͼ��ʾ��
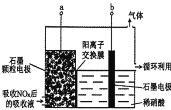
��ͼ��bӦ���ӵ�Դ��_________ �������������������
�ڽ�ʯī�缫���óɿ���״��Ŀ��_____________________��
�������ĵ缫��ӦʽΪ___________________��