��Ŀ����
��֪BaCO3��BaSO4�ܶȻ������ֱ�ΪKsp=5.1��10-9 mol2��L-2,Ksp=1.1��10-10 mol2��L-2,��BaSO4��������,BaCO3���������͵���Ҫ������(����)
| A��BaSO4���ܶȻ�������BaCO3��С |
| B��Sû�ж���,C�ж��� |
| C��BaSO4���ܶȴ� |
| D��θ������C���,����CO2��H2O,ʹBaCO3���ܽ�ƽ�������ƶ�,Ba2+Ũ������,�������Ba2+�ж� |
D
BaSO4������������Ϊ���ᱵ�������ᡣ��BaCO3�����ᷴӦ:BaCO3+2H+ Ba2++H2O+CO2��,���ɵ�Ba2+��������,������ؽ������ж�������D��ȷ��
Ba2++H2O+CO2��,���ɵ�Ba2+��������,������ؽ������ж�������D��ȷ��
 Ba2++H2O+CO2��,���ɵ�Ba2+��������,������ؽ������ж�������D��ȷ��
Ba2++H2O+CO2��,���ɵ�Ba2+��������,������ؽ������ж�������D��ȷ��
��ϰ��ϵ�д�
 Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д� Сѧͬ�����������ܾ�ϵ�д�
Сѧͬ�����������ܾ�ϵ�д�
�����Ŀ
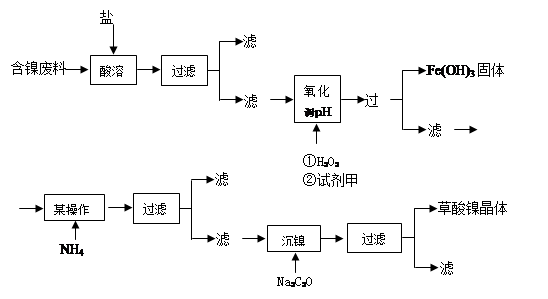
 LaNi5+6Ni(OH)2���õ�طŵ�ʱ��������Ӧ�� ��
LaNi5+6Ni(OH)2���õ�طŵ�ʱ��������Ӧ�� ��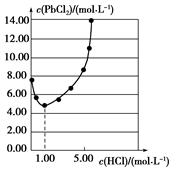
 Al3��(aq)��3OH��(aq)��ʾ�����ܽ�ƽ�⣬Al(OH)3
Al3��(aq)��3OH��(aq)��ʾ�����ܽ�ƽ�⣬Al(OH)3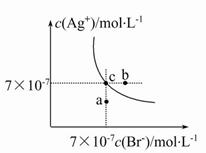
 AgBr(s)��Cl-(aq)ƽ�ⳣ��K��816
AgBr(s)��Cl-(aq)ƽ�ⳣ��K��816