��Ŀ����
��5�֣�������Ԫ�صĵ���X��Y��Z��ͨ��״���¾�Ϊ��̬����������ת����ϵ����Ӧ������ȥ����
��֪��a������˫ԭ�ӵ��ʷ����У�X��
�Ӻ����õ��Ӷ���Ŀ��ࡣb�����Ӻ�
10�����ӣ�����ʹʪ��ĺ�ɫʯ����ֽ
�������ҷ��Ӻ�18�����ӡ�
��1��X�Ľṹʽ�� �����ĵ���ʽ��
��2��ʵ���ҿ�����ͼ��ʾװ�ã�ȱ���ռ�װ�ã��г̶ֹ�װ����ȥ���Ʊ����ռ��ס�
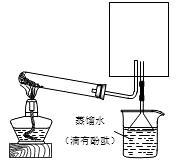
����ͼ�з����ڻ������ƿ�ռ�������װ�ü�ͼ��
���Թ��з�����Ӧ�Ļ�ѧ����ʽ�ǣ�
���ձ�����Һ����ɫ��Ϊ��ɫ����ԭ���ǣ����õ��뷽��ʽ��ʾ��

��֪��a������˫ԭ�ӵ��ʷ����У�X��
�Ӻ����õ��Ӷ���Ŀ��ࡣb�����Ӻ�
10�����ӣ�����ʹʪ��ĺ�ɫʯ����ֽ
�������ҷ��Ӻ�18�����ӡ�
��1��X�Ľṹʽ�� �����ĵ���ʽ��
��2��ʵ���ҿ�����ͼ��ʾװ�ã�ȱ���ռ�װ�ã��г̶ֹ�װ����ȥ���Ʊ����ռ��ס�

����ͼ�з����ڻ������ƿ�ռ�������װ�ü�ͼ��
���Թ��з�����Ӧ�Ļ�ѧ����ʽ�ǣ�
���ձ�����Һ����ɫ��Ϊ��ɫ����ԭ���ǣ����õ��뷽��ʽ��ʾ��
��1��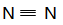
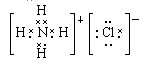
��2���� ��2NH4Cl+Ca��OH��2
��2NH4Cl+Ca��OH��2 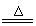 CaCl2+2NH3��+2H2O
CaCl2+2NH3��+2H2O
��NH3��H2O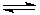 NH4++OH��
NH4++OH��
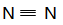
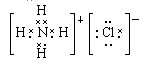
��2����
 ��2NH4Cl+Ca��OH��2
��2NH4Cl+Ca��OH��2  CaCl2+2NH3��+2H2O
CaCl2+2NH3��+2H2O ��NH3��H2O
 NH4++OH��
NH4++OH�����Ӻ�10�����ӣ�����ʹʪ��ĺ�ɫʯ����ֽ������˵�����ǰ���������ΪX��
�Ӻ����õ��Ӷ���Ŀ��࣬����X�ǵ�������Y���������ҷ��Ӻ�18�����ӣ��ҺͰ����������Ϸ�Ӧ�����������Ȼ��⣬��Z��������
��1�������к��е��������������Ȼ�泥��������Ӽ��ͼ��Լ����������ӻ����
��2���������ܶ�С�ڿ����ģ��Ҽ�������ˮ�������������ſ������ռ���������������ˮ����һˮ�ϰ����ܵ����OH������Һ�Լ��ԡ�
�Ӻ����õ��Ӷ���Ŀ��࣬����X�ǵ�������Y���������ҷ��Ӻ�18�����ӣ��ҺͰ����������Ϸ�Ӧ�����������Ȼ��⣬��Z��������
��1�������к��е��������������Ȼ�泥��������Ӽ��ͼ��Լ����������ӻ����
��2���������ܶ�С�ڿ����ģ��Ҽ�������ˮ�������������ſ������ռ���������������ˮ����һˮ�ϰ����ܵ����OH������Һ�Լ��ԡ�

��ϰ��ϵ�д�
�����Ŀ
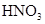 ��
��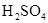 ���ʵ���Ũ�ȷֱ�Ϊ
���ʵ���Ũ�ȷֱ�Ϊ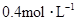 ��
��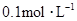 �������Һ�м���1.92gͭ�ۣ����ȣ�����ַ�Ӧ��������Һ��
�������Һ�м���1.92gͭ�ۣ����ȣ�����ַ�Ӧ��������Һ�� ���ʵ���Ũ�ȣ�
���ʵ���Ũ�ȣ�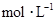 ��Ϊ������
��Ϊ������ N2��3H2O��3Cu����ʾ��ͼ�е�װ�ÿ���ʵ�ָ÷�Ӧ���ش��������⣺
N2��3H2O��3Cu����ʾ��ͼ�е�װ�ÿ���ʵ�ָ÷�Ӧ���ش��������⣺
 5N2+6 H2O ����ѧ�о���ѧϰС���ͬѧ�ڼ�����Ա��ָ���£����������̣�̽����ͬ������NH3��ԭNO��Ӧ�Ĵ����ܡ�
5N2+6 H2O ����ѧ�о���ѧϰС���ͬѧ�ڼ�����Ա��ָ���£����������̣�̽����ͬ������NH3��ԭNO��Ӧ�Ĵ����ܡ� ����������ʵ����������ͬ���ڴ���Ӧ����װ�ز�ͬ�Ĵ�������������Ӧ��Ļ�����壬ͨ��һ��������з�̪��ϡ������Һ����Һ�������Ũ�Ⱦ���ͬ����
����������ʵ����������ͬ���ڴ���Ӧ����װ�ز�ͬ�Ĵ�������������Ӧ��Ļ�����壬ͨ��һ��������з�̪��ϡ������Һ����Һ�������Ũ�Ⱦ���ͬ����