��Ŀ����
ˮ����������������������Ӧ�ù㷺��
��1�����н϶� ���ӵ�ˮ��ΪӲˮ��Ӳˮ���Ⱥ����̼���γ��������ӷ���ʽΪ ��д��һ�����ɣ���
��2����RH�������ӽ�����֬��ROH�������ӽ�����֬������������ȻӲˮ��Ӧ����ʹӲˮͨ�� ���RH����ROH���������ӽ�����֬��ԭ���� ��
��3��ͨ��ʩ��һ��ѹ��ʹˮ����ͨ����Ĥ��������ӻ����ӽ������Ӷ���ô���ˮ�ķ�����Ϊ ��������������ˮʱ��ʹ����ͨ����Ĥ���ƶ����� ��
��4����������ˮ�Ĵ���ʱ��������еķ����Dzⶨˮ�� ��
��5��Ӳ��Ϊ1���ˮ��ָÿ��ˮ��10mg CaO����֮�൱�����ʣ���7.1mg MgO������ij��Ȼˮ��c��Ca2+��=1.2��10-3mol/L��c��Mg2+��=6��10-4mol/L�����ˮ��Ӳ��Ϊ ��
��1�����н϶�
��2����RH�������ӽ�����֬��ROH�������ӽ�����֬������������ȻӲˮ��Ӧ����ʹӲˮͨ��
��3��ͨ��ʩ��һ��ѹ��ʹˮ����ͨ����Ĥ��������ӻ����ӽ������Ӷ���ô���ˮ�ķ�����Ϊ
��4����������ˮ�Ĵ���ʱ��������еķ����Dzⶨˮ��
��5��Ӳ��Ϊ1���ˮ��ָÿ��ˮ��10mg CaO����֮�൱�����ʣ���7.1mg MgO������ij��Ȼˮ��c��Ca2+��=1.2��10-3mol/L��c��Mg2+��=6��10-4mol/L�����ˮ��Ӳ��Ϊ
���㣺���ʵķ��롢�ᴿ�Ļ�������ѡ����Ӧ��,���ܵ���ʵ��ܽ�ƽ�⼰����ת���ı���
ר�⣺ʵ�������
��������1�����ơ�þ���ӽ϶��ˮΪӲˮ��Ӳˮ���Ⱥ����̼��Ƴ�����
��2��������ȻӲˮʱ��RH�������ӽ�����֬�ɽ���Ӳˮ�е�Ca2+��Mg2+���ӣ�����������Mg��OH��2�ȳ�����Ӱ����֬����Ч����
��3����ҵ�����÷�����ͨ��ʩ��һ��ѹ��ʹˮ����ͨ����Ĥ��������ӻ����ӽ���������ô���ˮ�����õ���������ʹ��Ӧ������ͨ����Ĥ�ԴﵽӲˮ������Ч����
��4����Һ�ĵ�������ȡ������Һ�е�����Ũ�ȴ�С����������ˮ�Ĵ���ʱ����ķ����Dzⶨˮ�ĵ����ʣ�
��5��Ӳ��Ϊ1���ˮ��ָÿ��ˮ��10mgCaO����֮�൱�����ʣ���7.1mgMgO������ˮ�е�Ca2+��Mg2+���������CaO����������õ���
��2��������ȻӲˮʱ��RH�������ӽ�����֬�ɽ���Ӳˮ�е�Ca2+��Mg2+���ӣ�����������Mg��OH��2�ȳ�����Ӱ����֬����Ч����
��3����ҵ�����÷�����ͨ��ʩ��һ��ѹ��ʹˮ����ͨ����Ĥ��������ӻ����ӽ���������ô���ˮ�����õ���������ʹ��Ӧ������ͨ����Ĥ�ԴﵽӲˮ������Ч����
��4����Һ�ĵ�������ȡ������Һ�е�����Ũ�ȴ�С����������ˮ�Ĵ���ʱ����ķ����Dzⶨˮ�ĵ����ʣ�
��5��Ӳ��Ϊ1���ˮ��ָÿ��ˮ��10mgCaO����֮�൱�����ʣ���7.1mgMgO������ˮ�е�Ca2+��Mg2+���������CaO����������õ���
���
�⣺��1��ͨ����ˮ��Ca2+��Mg2+�Ķ��٣�����Ȼˮ��ΪӲˮ����ˮ�����н϶�Ca2+��Mg2+��ˮ����Ӳˮ�����ˮ��Ӳ������Ca��HCO3��2��Mg��HCO3��2������ģ�����Ӳ�Ƚ�����ʱӲ�ȣ�������ʱӲ�ȵ�ˮ�����ü��ȵķ�����������������ʽΪCa2++2HCO3-
CaCO3��+CO2��+H2O��Mg2++2HCO3-
MgCO3��+CO2��+H2O��
�ʴ�Ϊ��Ca2+��Mg2+��Ca2++2HCO3-
CaCO3��+CO2��+H2O��Mg2++2HCO3-
MgCO3��+CO2��+H2O��
��2�����ˮ��Ӳ�����ɸƺ�þ�������λ��Ȼ��������ģ�����Ӳ�Ƚ�������Ӳ�ȣ���������Ӳ�ȵ�ˮ���Բ������ӽ������������������ӽ���������������ˮ�е�Ca2+��Mg2+�������ӽ������ã�ʹˮ�õ����������Ӳˮ��ͨ��ROH�������ӽ�����֬ʱ���ܲ���Mg��OH��2�ȳ�����Ӱ�콻��Ч����������ͨ��RH�������ӽ�����֬��
�ʴ�Ϊ��RH����ͨ�������ӽ�����֬��������Mg��OH��2�ȳ�����Ӱ����֬����Ч����
��3������������һ���������ӽ���Ĥ���к�ˮ�����ķ���������ӵ糡�������£�ˮ��Һ�����������ӻ�ֱ��������ƶ���������м����һ�ֽ���Ĥ���Ϳ��ܴﵽ����Ũ����Ŀ�ģ����Ե�����������ˮʱ��ʹ����ͨ����Ĥ���ƶ����ǵ��Ʋ��糡�����ʴ�Ϊ�������������Ʋ��糡������
��4����Ϊˮ�ĵ���̶ȼ�С�����Դ�ˮ�Ǽ���������ģ����Ҫ��������ˮ�Ĵ���ʱ��������еķ����Dzⶨˮ�ĵ絼�ʻ�����ʣ��ʴ�Ϊ���絼�ʣ�������ʣ���
��5��ij��Ȼˮ��c��Ca2+��=1.2��10-3mol?L-1��c��Mg2+��=6��10-4mol?L-1��Ӳ��Ϊ1���ˮ��ָÿ��ˮ��10mgCaO����֮�൱�����ʣ���7.1mgMgO����1Lˮ�и��������ʵ���=1.2��10-3mol���൱��CaO����=1.2��10-3mol��56g/mol=67.2mg��1Lˮ��þ�������ʵ���=6��10-4mol���൱������þ����6��10-4mol��40g/mol=24mg������ˮ��Ӳ��=
+
=10.1�㣬�ʴ�Ϊ��10.1�㣮
| ||
| ||
�ʴ�Ϊ��Ca2+��Mg2+��Ca2++2HCO3-
| ||
| ||
��2�����ˮ��Ӳ�����ɸƺ�þ�������λ��Ȼ��������ģ�����Ӳ�Ƚ�������Ӳ�ȣ���������Ӳ�ȵ�ˮ���Բ������ӽ������������������ӽ���������������ˮ�е�Ca2+��Mg2+�������ӽ������ã�ʹˮ�õ����������Ӳˮ��ͨ��ROH�������ӽ�����֬ʱ���ܲ���Mg��OH��2�ȳ�����Ӱ�콻��Ч����������ͨ��RH�������ӽ�����֬��
�ʴ�Ϊ��RH����ͨ�������ӽ�����֬��������Mg��OH��2�ȳ�����Ӱ����֬����Ч����
��3������������һ���������ӽ���Ĥ���к�ˮ�����ķ���������ӵ糡�������£�ˮ��Һ�����������ӻ�ֱ��������ƶ���������м����һ�ֽ���Ĥ���Ϳ��ܴﵽ����Ũ����Ŀ�ģ����Ե�����������ˮʱ��ʹ����ͨ����Ĥ���ƶ����ǵ��Ʋ��糡�����ʴ�Ϊ�������������Ʋ��糡������
��4����Ϊˮ�ĵ���̶ȼ�С�����Դ�ˮ�Ǽ���������ģ����Ҫ��������ˮ�Ĵ���ʱ��������еķ����Dzⶨˮ�ĵ絼�ʻ�����ʣ��ʴ�Ϊ���絼�ʣ�������ʣ���
��5��ij��Ȼˮ��c��Ca2+��=1.2��10-3mol?L-1��c��Mg2+��=6��10-4mol?L-1��Ӳ��Ϊ1���ˮ��ָÿ��ˮ��10mgCaO����֮�൱�����ʣ���7.1mgMgO����1Lˮ�и��������ʵ���=1.2��10-3mol���൱��CaO����=1.2��10-3mol��56g/mol=67.2mg��1Lˮ��þ�������ʵ���=6��10-4mol���൱������þ����6��10-4mol��40g/mol=24mg������ˮ��Ӳ��=
| 67.2mg |
| 10mg |
| 24mg |
| 7.1mg |
���������⿼��Ӳˮ������ԭ��������ʱע��Ӳˮ����Ҫ���ӳɷ��Լ����ʵ����ʣ���5���м���Ϊ�����״��㣬ע����ػ���֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
�������ڻ�ѧ���ʵ��� ��������
| A���ܽ��� | B���ӷ��� |
| C��������� | D�������� |
��ͬ�¶��£���ˮ�зֱ�����������ʣ�����KW��ʾˮ�����ӻ�������ʾˮ�ĵ���ȣ���ʹ
��ֵ������ǣ�������
| �� |
| KW |
| A��NaOH |
| B��NaHSO4 |
| C��NH4NO3 |
| D��KCl |
����������ȷ���ǣ�������
A��25��ʱ����0.1mol/L��ˮ��Һ��
| ||
B��25��ʱ����0.1mol/L������Һ��ˮϡ�ͣ������¶Ȳ��䣬ϡ������
| ||
| C��0.1mol/L��Na2CO3��Һ�У�c��OH-��-c��H+��=c��HCO3-��+2c��H2CO3�� | ||
| D��Ũ�Ⱦ�Ϊ0.1mol/L�İ�ˮ��Һ��NH4Cl��Һ�������Ϻ�����Һ�ʼ��ԣ���������Һ�У�c��NH4+����c��NH3?H2O����c��Cl-����c��OH-����c��H+�� |
�����������ȷ�Ӧ���ǣ�������
| A��CaO+H2O�TCa��OH��2 | ||||
B��C+H2O
| ||||
| C��NaOH+HCl�TNaCl+H2O | ||||
D��2Mg+CO2
|
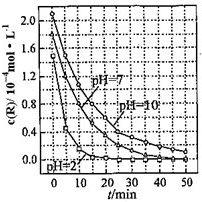 һ�������£���Һ������Զ�TiO2���ȼ��R���ⷴӦ��Ӱ����ͼ��ʾ����ش��������⣺
һ�������£���Һ������Զ�TiO2���ȼ��R���ⷴӦ��Ӱ����ͼ��ʾ����ش��������⣺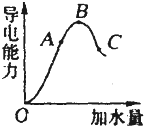 ��һ���¶��£��������ˮϡ�����У���Һ�ĵ���������ͼ��ʾ����ش�
��һ���¶��£��������ˮϡ�����У���Һ�ĵ���������ͼ��ʾ����ش�