��Ŀ����
��ҵ������п����Ҫ�ɷ�ΪZnS��������Fe2O3�����ʣ�Ϊԭ������ZnSO4��7H2O�Ĺ����������£�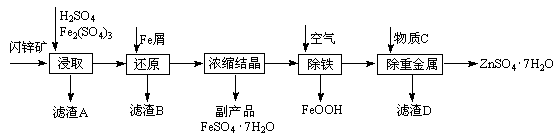
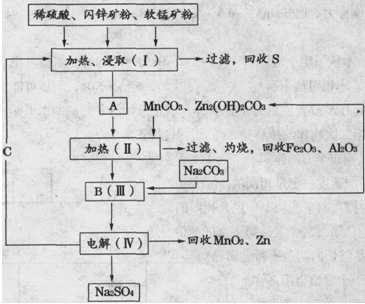
��1��������A�пɻ��һ�ֵ���ɫ�ǽ������ʵĸ���Ʒ���仯ѧʽΪ ��
��2����ȡ������Fe2(SO4)3�������� ����ȡʱFe2(SO4)3��ZnS������Ӧ�Ļ�ѧ����ʽΪ ��
��3���������̿�����Һ��pH��5.4���ң��÷�Ӧ�����ӷ���ʽΪ ���ù����ڿ�����ڴ������һ��������ԡ��ͷ��װ�ã���Ŀ���� ��
��4���û������ؽ���������������CΪ ��
��5������п���ܽ�����¶�֮��Ĺ�ϵ���±���
| �¶�/�� | 0 | 20 | 40 | 60 | 80 | 100 |
| �ܽ��/g | 41.8 | 54.1 | 70.4 | 74.8 | 67.2 | 60.5 |
�ӳ��ؽ����������п��Һ�л������п�����ʵ�����Ϊ �� �����ˡ����
��1��S��2�֣�
��2������������2�֣� Fe2(SO4)3��ZnS��ZnSO4��2FeSO4��S��3�֣�
��3��4Fe2+��O2��6H2O��4FeOOH��8H+��3�֣�
�����������Һ�ĽӴ�������ӿ췴Ӧ���ʣ�2�֣�
��4��Zn��п����2�֣�
��5��60������������Ũ�������½ᾧ��2�֣�
���������������1����������ת����ϵ�����жϣ�������������������������ܽ�������п�����ӣ�������A�пɻ��һ�ֵ���ɫ�ǽ������ʵĸ���ƷΪ��������Ϊ�ʴ�Ϊ��S��
��2���������̷�Ӧ���������ʿ�֪��������������������������п�� ���ӣ����ɵ�����Ӧ�Ļ�ѧ����ʽΪ��Fe2��SO4��3+ZnS=ZnSO4+2FeSO4+S���ʴ�Ϊ������������ Fe2��SO4��3+ZnS=ZnSO4+2FeSO4+S��
��3���������̿�����Һ��pH��5.4���ң����ÿ�������������������������FeOOH����Ӧ�����ַ���ʽΪ��4Fe2++O2+6H2O=4FeOOH+8H+���ڿ�����ڴ������һ��������ԡ��ͷ��װ����Ϊ������������ĽӴ��������Ӧ���ʣ��ʴ�Ϊ��4Fe2++O2+6H2O=4FeOOH+8H+�������������Һ�ĽӴ�������ӿ췴Ӧ���ʣ�
��4���Ʊ�ʵ�鷽������ȡZnSO4?7H2O�����ݳ��Ӳ��������µ����ʣ�Cd�Ľ�����Խ���Zn��Fe֮�䣬���Լ���п�����û��ӣ��ʴ�Ϊ��Zn��
��5������ͼ��������п���ܽ�����¶�֮��Ĺ�ϵ���������º����ܽ�ȶ��������ֻ����60��C����п���ܽ�����74.8g��ͨ����������Ũ������ȴ�ᾧ�����̶ȵõ����壬
�ʴ�Ϊ��60������������Ũ�������½ᾧ����
���㣺���������Ʊ����������̷����ͷ�Ӧ�жϣ�ʵ��������ϢӦ�õ����֪ʶ��

���������Ҫ�ɷֿɱ�ʾΪFeO��Cr2O3��������MgO��Al2O3��Fe2O3�����ʣ��������Ը�����Ϊԭ���Ʊ��ظ���أ�K2Cr2O7��������ͼ��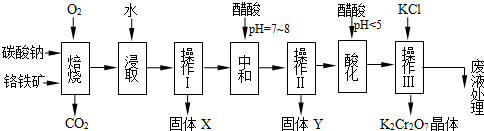
��֪����4FeO��Cr2O3+ 8Na2CO3+ 7O2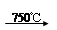 8Na2CrO4 + 2 Fe2O3 + 8CO2����
8Na2CrO4 + 2 Fe2O3 + 8CO2����
��Na2CO3 + Al2O3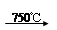 2NaAlO2 + CO2������ Cr2O72��+ H2O
2NaAlO2 + CO2������ Cr2O72��+ H2O 2CrO42�� + 2H+
2CrO42�� + 2H+
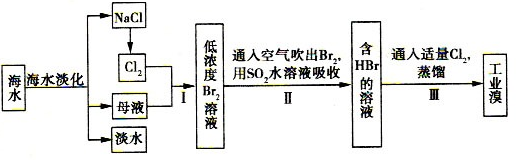
��1������X����Ҫ����_________����д��ѧʽ����Ҫ����ữ��������Һ��pH�Ƿ����4.5��Ӧ��ʹ��__________����д�������Լ����ƣ���
��2���ữ�����ô��������ҺpH��5����Ŀ����_________________________________��
| ���� | �ܽ��/(g/100gˮ) | ||
| 0��C | 40��C | 80��C | |
| KCl | 28 | 40.1 | 51.3 |
| NaCl | 35.7 | 36.4 | 38 |
| K2Cr2O7 | 4.7 | 26.3 | 73 |
| Na2Cr2O7 | 163 | 215 | 376 |
��3���������жಽ��ɣ����K2Cr2O7����IJ��������ǣ�����KCl���塢����Ũ���� ��
���ˡ�_______�����
��4���ұ���������ʵ��ܽ�����ݣ�����������Ӧ�Ļ�ѧ����ʽ�ǣ�Na2Cr2O7+2KCl��K2Cr2O7��+2NaCl���÷�Ӧ����Һ���ܷ�����������_______________��
��5��������Һ�й���������ʹCr2O72-������ɫ�Ĺ���������CrO5���ӽṹΪ
 ), �÷�Ӧ����������Cr2O72-�Ĵ��ڡ�д����Ӧ�����ӷ���ʽ�� ��
), �÷�Ӧ����������Cr2O72-�Ĵ��ڡ�д����Ӧ�����ӷ���ʽ�� ���÷�Ӧ (����ڡ������ڡ�)������ԭ��Ӧ��
��6����ȡ�ظ��������2.500g���250mL��Һ,ȡ��25mL�����ƿ��,����10mL2mol/ L H2SO4�������⻯�أ����Ļ�ԭ����ΪCr3+)�����ڰ���5min��Ȼ�����100mLˮ������3mL����ָʾ������0.1200mol/LNa2S2O3����Һ�ζ���I2+2S2O32����2I-+S4O62������
���жϴﵽ�ζ��յ�������� ��
����ʵ���й���ȥNa2S2O3����Һ40.00mL�������ò�Ʒ���ظ���صĴ���Ϊ ��������3λ��Ч����, K2Cr2O7��Ħ������Ϊ294g/mol����

 MnO2+Zn+2H2SO4��
MnO2+Zn+2H2SO4��