��Ŀ����
18����̼���ƣ�Na2CO4����ϴ�ӡ�ӡȾ����֯����ֽ��ҽҩ�����������д���Ӧ�ã�ij��ѧ�о���ѧϰС����Ʋ����������ʵ�飮�ⶨ��̼������Ʒ�����е����ʲ�����ˮ�Ҳ����ᷴӦ���Ĵ��ȣ�ʵ��ԭ��2Na2CO4+4HCl�T4NaCl+2CO2��+O2��+2H2O
ʵ��װ��

����������
��ͬѧ��Ϊ�ⶨ������O2��������ɲⶨ��Ʒ�Ĵ��ȣ�
��1��װ�âڵ������dz�ȥ������̼���Ȼ�������
��2��װ�â۵������ǣ���֤������̼�Ƿ���
��3����ȡ������Ʒ������Ũ�����ַ�Ӧ���ռ��������������Ϊ0.25L����֪��ʵ���������������ܶ�Ϊ1.28g/L�������ȡ����Ʒ����Ӧ����2.44g
����ͬѧ��Ϊ�ⶨ������CO2������������Ӧǰ��װ�âڵ�������Ϳɲⶨ��Ʒ�Ĵ��ȣ�����������ʵ��װ�ò��CO2��������������Ʒ�Ĵ��ȣ������ƫ�ߣ�����Ϊԭ�������ABC��������ţ�
A��Ũ�����ӷ����������Ȼ������屻װ�â�����
B��������̼�����ݳ�ʱ������ˮ������װ�â���������������
C��װ�â١����ڿ����еĶ�����̼��װ�â�����
��ͬѧ�����һ���·������õ��˴�ҵ�һ���Ͽɣ���ȡ��Ʒ12.5g���μ�����Ũ��������Ʒ��Ӧ��ȫ���ٽ���ƿ�����е�Һ���������ᾧ����ɣ��Ƶ��Ȼ��ƹ���11.7g�������Ʒ�Ĵ���Ϊ97.6%��
���� ��1������ʵ��ԭ��2Na2CO4+4HCl�T4NaCl+2CO2��+O2��+2H2O��Ҫ�ⶨ���������������ȳ�ȥ������̼���Ȼ�������ȣ��ٸ���ʵ��װ��ͼ���ɵó��𰸣�
��2��������̼��ʹ����ʯ��ˮ����ǣ������ô�������������̼��
��3��������������������ٸ��ݻ�ѧ����ʽ���������̼���Ƶ�������ϵ�������б���������Ʒ�еĹ�̼���Ƶ���������ô��Ʒ��������Ӧ�ô������������
�������Ͽ��Ը������ɶ�����̼��������Ʒ���ȣ�����ʵ��������ͨ��ʵ���ô����Ķ�����̼����������Ϊ��Ӧǰ��װ�âڵ��������л������Ȼ����ˮ����������������������⣻
���ݻ�ѧ����ʽ���Ȼ������̼���Ƶ�������ϵ�������б��������Ʒ�еĹ�̼���Ƶ�������
��� �⣺��1��װ�â���Ũ�������Ƶ������dz�ȥ������̼���Ȼ������壬
�ʴ�Ϊ����ȥ������̼���Ȼ������壻
��2��װ�â��г���ʯ��ˮ����������֤������̼�Ƿ�����
�ʴ�Ϊ����֤������̼�Ƿ�����
��3�����ռ��������������Ϊ0.25L����Ҫ��̼���Ƶ�����Ϊx��
2Na2CO4+4HCl�T4NaCl+2CO2��+O2��+2H2O
244 32
x 0.25L��1.28g/L
x=2.44g��
�ʴ�Ϊ��2.44��
��ö�����̼�����ƫ��ԭ������У�Ũ�����ӷ����������Ȼ������屻װ�â����գ�������̼�����ݳ�ʱ������ˮ������װ�â��������������գ�װ�â١����ڿ����еĶ�����̼��װ�â����գ���ѡABC��
�ʴ�Ϊ��ABC��
������Ʒ�й�̼���Ƶ�����Ϊy��
2Na2CO4+4HCl�T4NaCl+2CO2��+O2��+2H2O
244 234
y 11.7g
y=12.2g
����Ʒ�Ĵ���Ϊ��$\frac{12.2g}{12.5g}$��100%=97.6%
�ʴ�Ϊ��97.6%��
���� ���⿼��ʵ��װ�úͻ��������Ʒ���ȵļ��㣮�йػ�ѧ����ʽ�ļ��㣬Ҫע����뷽��ʽ����ı�����ʵ�ʲμӷ�Ӧ��ʵ�����ɵ���������������ͻ����ʵ������ƫ���ƫС����Ŀ�Ѷ��еȣ�

| A�� | ʵ��ʽCH2 | B�� | �ṹ��ʽCH2=CHCH3 | ||
| C�� | ���ģ��  | D�� | �ۺϺ�Ľṹ��ʽ |
| A�� | ��ϩ�Ľṹ��ʽΪCH2CH2 | |
| B�� | ����������1molCH4��3molCl2��Ӧ���Ƶ�1molCHCl3 | |
| C�� |  Ϊ�����ӵı���ģ�� Ϊ�����ӵı���ģ�� | |
| D�� |  ����̼ԭ���п��ܶ���ͬһ��ƽ���� ����̼ԭ���п��ܶ���ͬһ��ƽ���� |
| A�� | CO ȼ�յ��Ȼ�ѧ����ʽΪ 2CO��g��+O2��g���T2CO2 ��g����H=+282.8kJ/mol | |
| B�� | H2ȼ�յ��Ȼ�ѧ����ʽΪ 2H2��g��+O2��g���T2H2O��g����H=-571.6kJ/mol | |
| C�� | ȼ��ǰ�Ļ�������У�H2���������Ϊ40% | |
| D�� | �������ȼ�պ��������Ĺ������Ʒ�Ӧ��ת�Ƶ���2mol |
 �ס��ҡ����������ת����ϵ��ͼ��ʾ�����ַ�Ӧ�P��Ӧ��������ȥ����ͷ��ʾһ��ת���������и��������У���������ͼʾת����ϵ���ǣ�������
�ס��ҡ����������ת����ϵ��ͼ��ʾ�����ַ�Ӧ�P��Ӧ��������ȥ����ͷ��ʾһ��ת���������и��������У���������ͼʾת����ϵ���ǣ�������| ѡ�� | �� | �� | �� | �� |
| A | CH��CH | CH2=CH2 | CH2BrCH2Br | CH2OHCH2OH |
| B | NaAlO2 | Al��OH��3 | Al2O3 | Al |
| C | CH3CH2OH | CH3CHO | CH3COOH | CH3COOC2H5 |
| D | SiO2 | H2SiO3 | Na2SiO3 | Si |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ʵ������ͭ��Ũ���ᷴӦ��ȡ����ͭ | |
| B�� | ʵ������ͭ��ϡ���ᷴӦ��ȡһ������ | |
| C�� | ��ҵ�����õ�����������ȡ���� | |
| D�� | ��ҵ���ý�̿��ԭ������������ȡ�� |
 ��
��


 +2NaOH$��_{��}^{H_{2}O}$
+2NaOH$��_{��}^{H_{2}O}$ +2NaBr��
+2NaBr��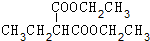 +��CH3��2CHCH2CH2Br+CH3CH2ONa��
+��CH3��2CHCH2CH2Br+CH3CH2ONa�� +CH3CH2OH+NaBr��
+CH3CH2OH+NaBr�� ��
�� ��
��