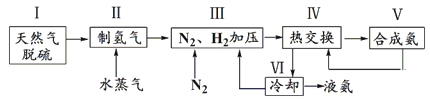
��Ŀ����
����Ŀ��Ŀǰ��ȼú�����ǿ��й������о�����Ҫ����֮һ����Ҫ���������¼��֣�
��ʯ�ҷ�ԭ��Ϊ��2CaO(s)+2SO2(g)+O2(g)2CaSO4(s)
��1��T1��ʱ����10L�����ܱ������м���3molCaO(s)����ͨ��2molSO2(g)��1molO2(g)����������Ӧ��2minʱ��ƽ�⣬��ʱCaSO4(s)Ϊ1.8mol��0~2min�ڣ���SO2(g)��ʾ�ĸ÷�Ӧ������v(SO2)=__________________________��
��������
����̿���������ǻ��ڻ������õ����������ͻ�ѧ�������ã��䷴Ӧ����Ϊ��
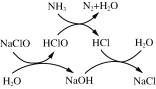
O2+2C��2C��O��SO2+C��C��SO2��C��SO2+C��O��C��SO3+C��
C��SO3+H2O��C��H2SO4��C��H2SO4��H2SO4+C��
��2�������������ܵĻ�ѧ��Ӧ����ʽΪ__________________________________________��
��3������̿�ڷ�Ӧ���������������ı���_____________����ѡ����ĸ����
A����Ӧ���� B����Ӧ�� C����Ӧ�ʱ� D����Ӧ·�� E����Ӧ���
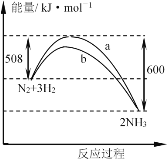
��CO��ԭ��ԭ��Ϊ2CO(g)+SO2(g)S(g)+2CO2(g) ��H
��֪��S(g)+O2(g)=SO2(g) ��H1=-574.0kJ��mol-1��COȼ����Ϊ283.0kJ��mol-1��
��4����ʼ�¶�ΪT2��ʱ���ֱ��������ݻ���Ϊ10L�ľ����ܱ������У�������Ӧ2CO(g)+SO2(g)S(g)+2CO2(g)���������������±���ʾ��
���� | ��ʼʱ���ʵ���/mol | ƽ��ʱCO2(g)�����ʵ���/mol | |||
CO(g) | SO2(g) | S(g) | CO2(g) | ||
�� | 1 | 0.5 | 0.5 | 0 | A |
�� | 1 | 0.5 | 0 | 0 | 0.8 |
�� | 2 | 1 | 0 | 0 | b |
�������Ҵﵽƽ����������������䣬�������г���CO(g)�� SO2(g)��S(g)��CO2(g)��1mol����ʱv(��)__________ v(��)�������������������=��)��
�������ж���ȷ����________________����ѡ����ĸ����
A��b=1.6 B��ƽ�ⳣ���ף��� C���ﵽƽ���ʱ�䣺������
���𰸡�0.09mol��L-1��min-1 2SO2 + O2 + 2H2O = 2H2SO4 A��D��E �� BC
��������
I��(1)���ݷ�Ӧ2CaO(s)+2SO2(g)+O2(g)2CaSO4(s)��֪����1.8molCaSO4��Ҫ����1.8mol SO2����2min��SO2Ũ�ȱ仯��=![]() =0.18mol/L����SO2(g)��ʾ�ĸ÷�Ӧ����=
=0.18mol/L����SO2(g)��ʾ�ĸ÷�Ӧ����=![]() =0.09mol/(L��min)��
=0.09mol/(L��min)��
II��(2)��֪��O2 +2C��2C��O����SO2 +C��C��SO2����C��SO2 + C��O��C��SO3 + C����C��SO3+H2O�� C��H2SO4����C��H2SO4��H2SO4 + C����[��![]() +��+��+��+��]
+��+��+��+��]![]() 2��2SO2 + O2 + 2H2O
2��2SO2 + O2 + 2H2O![]() 2H2SO4��
2H2SO4��
(3)�����ܹ���С��Ӧ�Ļ�ܣ��Ӷ��ı䷴Ӧ���ʣ��������뷴Ӧ���ı䷴Ӧ·�����ɴ˽��
III��(4)��֪��COȼ����Ϊ283.0 kJ��mol-1�����Ȼ�ѧ����ʽ����2CO(g)+O2(g)=2CO2(g), ��H=-566.0kJ/mol����S(g)+O2(g)=SO2(g),��H1=-574.0 kJ��mol-1�����ݸ�˹���ɽ��٣��ڵ�2CO(g)+SO2(g)S(g)+2CO2(g) ,��H=-566.0kJ/mol-(-574.0kJ/mol)=+8kJ/mol����2CO(g)+SO2(g)S(g)+2CO2(g) ,��H=+8kJ/mol������Ϊ�������¶Ȳ��䣬����ƽ�ⳣ�����䣬���������г���CO(g)�� SO2(g)��S(g)��CO2(g)��1mol��������Ũ����Q=![]() ����ƽ�ⳣ���ȽϽ�𡣢�A.�ٶ�����������������ͬ�¶Ⱥ����������������ʼ���ʵ����������ҳ�ʼ���ʵ�����2��������������Ӧ����������ҷ�Ӧ��Ч�ڼ�ѹ���ٿ���ʵ�����ھ��������£��Ƚ����������¶��������ҵ��¶ȸߵ�������B.�ٶ������ס��Ҵ��ں��º����£��ӱ����ʼͶ�Ͽ��������״ﵽ��ƽ���൱����������ƽ�����ټ���0.5molS(g)��������ƽ�����淴Ӧ�����ƶ�����������Ӧ���յ��������������٣��������ס���ʵ�ʴ��ھ��������£��ٱȽ��������������ҵ��¶ȸߵͣ�����ƽ�ⳣ�����¶��йط�����C.�ٶ��ں��º����£��ҡ����������ﵽ��ƽ�⻥Ϊ��Чƽ�⣬���������յ������������ҵ�2�����������и���ֵ�Ũ�����������ж�Ӧ���Ũ�ȵ�2�������������������Ҷ����յ�����=
����ƽ�ⳣ���ȽϽ�𡣢�A.�ٶ�����������������ͬ�¶Ⱥ����������������ʼ���ʵ����������ҳ�ʼ���ʵ�����2��������������Ӧ����������ҷ�Ӧ��Ч�ڼ�ѹ���ٿ���ʵ�����ھ��������£��Ƚ����������¶��������ҵ��¶ȸߵ�������B.�ٶ������ס��Ҵ��ں��º����£��ӱ����ʼͶ�Ͽ��������״ﵽ��ƽ���൱����������ƽ�����ټ���0.5molS(g)��������ƽ�����淴Ӧ�����ƶ�����������Ӧ���յ��������������٣��������ס���ʵ�ʴ��ھ��������£��ٱȽ��������������ҵ��¶ȸߵͣ�����ƽ�ⳣ�����¶��йط�����C.�ٶ��ں��º����£��ҡ����������ﵽ��ƽ�⻥Ϊ��Чƽ�⣬���������յ������������ҵ�2�����������и���ֵ�Ũ�����������ж�Ӧ���Ũ�ȵ�2�������������������Ҷ����յ�����=![]() ��8kJ/mol��0.8mol=3.2kJ����ʵ�����ھ��������£���ʱŨ�ȶԷ�Ӧ���ʵ�Ӱ�쳬�����¶ȶԷ�Ӧ���ʵ�Ӱ�죬���������ķ�Ӧ���ʱ������ҵķ�Ӧ���ʴ�Ӧ����Խ��ﵽƽ���ʱ��ԽС���ɴ˷�����
��8kJ/mol��0.8mol=3.2kJ����ʵ�����ھ��������£���ʱŨ�ȶԷ�Ӧ���ʵ�Ӱ�쳬�����¶ȶԷ�Ӧ���ʵ�Ӱ�죬���������ķ�Ӧ���ʱ������ҵķ�Ӧ���ʴ�Ӧ����Խ��ﵽƽ���ʱ��ԽС���ɴ˷�����
I��(1)���ݷ�Ӧ2CaO(s)+2SO2(g)+O2(g)2CaSO4(s)��֪����1.8molCaSO4��Ҫ����1.8mol SO2����2min��SO2Ũ�ȱ仯��=![]() =0.18mol/L����SO2(g)��ʾ�ĸ÷�Ӧ����=
=0.18mol/L����SO2(g)��ʾ�ĸ÷�Ӧ����=![]() =0.09mol/(L��min)��
=0.09mol/(L��min)��
II��(2)��֪��O2 +2C��2C��O����SO2 +C��C��SO2����C��SO2 + C��O��C��SO3 + C����C��SO3+H2O�� C��H2SO4����C��H2SO4��H2SO4 + C����[��![]() +��+��+��+��]
+��+��+��+��]![]() 2��������2SO2 + O2 + 2H2O
2��������2SO2 + O2 + 2H2O![]() 2H2SO4
2H2SO4![]() 2H2SO4��
2H2SO4��
(3)�����ܹ���С��Ӧ�Ļ�ܣ������Ӱٷ������Ӷ��ı䷴Ӧ�������������뻯ѧ��Ӧ���ı䷴Ӧ·������������Ӱ��ƽ���ƶ������Բ��ı䷴Ӧ�ȣ��������ܸı䷴Ӧ�����������е��������ʲ��ı䷴Ӧ�ʱ䣬��ѡADE��
III��(4)��֪COȼ����Ϊ283.0 kJ��mol-1�����Ȼ�ѧ����ʽ����2CO(g)+O2(g)=2CO2(g), ��H=-566.0kJ/mol����S(g)+O2(g)=SO2(g),��H1=-574.0 kJ��mol-1�����ݸ�˹���ɽ��٣��ڵ�2CO(g)+SO2(g)S(g)+2CO2(g),��H=-566.0kJ/mol-(-574.0kJ/mol)=+8kJ/mol����2CO(g)+SO2(g)S(g)+2CO2(g) ,��H=+8kJ/mol��
�����ݱ������ݿ�֪�����Ҵﵽƽ��ʱ����0.8molCO2���ɷ���ʽ2CO(g)+SO2(g)S(g)+2CO2(g)������ϵ�����ƽ��ʱ��S(g)���ʵ���= 0.4mol��SO2���ʵ���=0.5mol-0.8mol��![]() =0.1mol��CO�����ʵ���=1mol-0.8mol=0.2mol�����¶���ƽ�ⳣ��K=
=0.1mol��CO�����ʵ���=1mol-0.8mol=0.2mol�����¶���ƽ�ⳣ��K=![]() =
= = 64�������¶Ȳ����ٳ���CO(g)�� SO2(g)��S(g)��CO2(g)��1mol����Ũ����Q=
= 64�������¶Ȳ����ٳ���CO(g)�� SO2(g)��S(g)��CO2(g)��1mol����Ũ����Q=![]() =
= =
=![]() <K=64��˵��ƽ��������Ӧ������У�����ʱv(��)>v(��)��
<K=64��˵��ƽ��������Ӧ������У�����ʱv(��)>v(��)��
��A.�ٶ�����������������ͬ�¶Ⱥ����������������ʼ���ʵ����������ҳ�ʼ���ʵ�����2��������������Ӧ����������ҷ�Ӧ��Ч�ڼ�ѹ������Ӧ2CO(g)+SO2(g)S(g)+2CO2(g) ,��H=+8kJ/molǰ�����������Ŀ���䣬�÷�Ӧ��ѹƽ�ⲻ�ƶ�����ƽ��ʱ��������CO2���ʵ�������������CO2���ʵ�����2����Ϊ1.6mol�����յ�����Ҳ�������ҵ�2�������ǣ�ʵ���Ǿ��������£������뻷���������������������������¶ȱ������ҵͣ����¸÷�Ӧ���淴Ӧ�����ƶ���������������������CO2������Ҫ���٣�����b<1.6��A�������B.�ٶ������ס��Ҵ��ں��º����£��ӱ����ʼͶ�Ͽ��������״ﵽ��ƽ���൱����������ƽ�����ټ���0.5molS(g)��������ƽ�����淴Ӧ�����ƶ�����������Ӧ���յ��������������٣����ǣ������ס���ʵ�ʴ��ھ��������£����������¶ȱ��������¶�Ҫ�ߣ�ƽ�ⳣ�����¶��йأ��ָ÷�Ӧ����ӦΪ���ȷ�Ӧ������ƽ��������Ӧ�����ƶ�������ƽ�ⳣ����>�ң�B����ȷ��C.����A�������֪�ٶ��ں��º��������ҡ����������ﵽ��ƽ�⻥Ϊ��Чƽ�⣬���������յ������������ҵ�2�����������и���ֵ�Ũ�����������ж�Ӧ���Ũ�ȵ�2�������������������Ҷ����յ�����=![]() ��8kJ/mol��0.8mol=3.2kJ����ʵ�����ھ��������£������������յ�3.2kJ�������������¶Ƚ��Ͳ��������������и���ֵ�Ũ���������ҵ�2������ʱŨ�ȶԷ�Ӧ���ʵ�Ӱ�쳬�����¶ȶԷ�Ӧ���ʵ�Ӱ�죬���������ķ�Ӧ���ʱ������ҵķ�Ӧ���ʴﵽƽ���ʱ���<�ң�C����ȷ����ѡBC��
��8kJ/mol��0.8mol=3.2kJ����ʵ�����ھ��������£������������յ�3.2kJ�������������¶Ƚ��Ͳ��������������и���ֵ�Ũ���������ҵ�2������ʱŨ�ȶԷ�Ӧ���ʵ�Ӱ�쳬�����¶ȶԷ�Ӧ���ʵ�Ӱ�죬���������ķ�Ӧ���ʱ������ҵķ�Ӧ���ʴﵽƽ���ʱ���<�ң�C����ȷ����ѡBC��

 �ǻۿ����ܾ�100�ֵ�Ԫ���ؼ��ϵ�д�
�ǻۿ����ܾ�100�ֵ�Ԫ���ؼ��ϵ�д� ��Ԫ������ĩ��ϵ�д�
��Ԫ������ĩ��ϵ�д�����Ŀ��������������(FeC2O4��xH2O)Ϊ����ɫ��ĩ��������ˮ������������Ӱ����������ҩ��ҵ��ij��ѧ��ȤС��������ʽ�������̽�����ش��������⣺
I.����̽��
ѡ�������Լ����ʵ�鷽��������±����ݡ�
�Լ�������KMnO4��Һ��K3[Fe(CN)6]��Һ
���� | ���� | ��������� |
(1)ȡ�������������������Թ��У�����2mLˮ������ | �е���ɫ�������ϲ���Һ��ɫ | ��������������ˮ |
(2)��������2mLϡ���ᣬ�� | ___________ | ���������������ᣬ��������ǿ�ڲ��� |
(3)����(2)������Һ�еμӼ���K3[Fe(CN)6]��Һ | ������ɫ���� | ___________ |
(4)��___________ | ��___________ | H2C2O4��C2O42�����л�ԭ�� |
��.����̽�����ζ�ʵ���x��ֵ
(5)�ζ�ǰ�����в�������ȷ˳����___________(����ĸ���)��
a.��0.1000mol/L������KMnO4��Һ��ϴ
b.��©����ϴ
c.�ž��ζ��ܼ�������ݲ�����Һ��
d.ʢװ0.1000mol/L������KMnO4��Һ
e.��ʼ��������¼Ϊ0.50mL
(6)��ȡng��Ʒ����������ϡ�����ܽ⣬�ò���(5)���ı�KMnO4��Һֱ�ӵζ�������жϵζ��յ�?_______________��
(7)�յ����Ϊ20.50mL���������ʵ���������x=___________(�ú�n�Ĵ���ʽ��ʾ��FeC2O4����Է�������Ϊ144)��