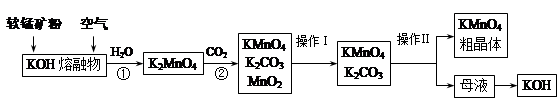��Ŀ����
�����������γɵĸ�����ʯ�ࣨCaSO4��2H2O��ת��Ϊ����ط��Ϻ��Ȼ���ˮ���ﴢ�Ȳ��ϣ����۴Ӿ���Ч�桢��Դ�ۺ����û��Ǵӻ��������Ƕȿ���������Ҫ���塣������ʯ��ת��Ϊ����غ��Ȼ��ƵĹ�������ʾ��ͼ��
��1�������������õ�ԭ�ϳ�CaSO4��2H2O��CaCO3��H2O�⣬����Ҫ ��ԭ�ϡ�
��2��д��ʯ������Һ�м���̼�����Һ������Ӧ�����ӷ���ʽ�� ��
��3�����ˢ����������Һ��(NH4)2SO4��Һ��������Һ�к���CO32���IJ��������ǣ�
_________________________________________________________________��
��4��������I����Һ�е�Ca2+Ũ�ȵ���1.0��10��5 mol��L��1ʱ����Ϊ��������ȫ����ҪʹCa2+������ȫ������Һ��CO32�����ӵ����ʵ���Ũ�Ȳ��õ��� ��
����֪��Ksp(CaCO3)=5.0��10��9��
��5��д�����������еĻ�ѧ����ʽ ��
��6���Ȼ��ƽᾧˮ���CaCl2��6H2O����Ŀǰ���õ������Ȳ��ϣ�ѡ��������� ��
a���۵�ϵͣ�29���ۻ��� b���ܵ��� c�������� d����
��13�֣� ��
��1��KCl NH3��2�֣���д1�����д1����1�֣�
��2��CaSO4��CO32�� CaCO3��SO42����2�֣����ӷ��Ŵ�����0�֣��õȺ�Ҳ���֣�
CaCO3��SO42����2�֣����ӷ��Ŵ�����0�֣��õȺ�Ҳ���֣�
��3���õι�ȡ������Һ���Թ��У��μ����ᣬ���������ݣ�֤����Һ�к���CO32��(3�֣�ÿ��1�֣���һ���1��)
��4��5.0��10��4mol/L��2�֣�û��д��λ��1�֣�
��6��a d��2�֣���1�����1����1�֣�
����

�����ܴﵽʵ��Ŀ�ĵ���



| A�������� | B����ˮ���ռ�NO2 | C��ʵ�����ư��� | D����ȥ��������HCl |
�����仯�������ִ���ҵ�ϵ�Ӧ�ù㷺�������ڵ�ƣ�������ë��Ƥ���������������ϣ�����ӡˢ��������ý���ȡ����������ϸ��������彡���кܴ�Σ����
��ij�������Ƹ﹤ҵ������Cr��III�������������ù������£������ȡҺ�н���������Ҫ��Cr3+�������Fe3����Al3+��Ca2+��Mg2+����
���������ӳ�����������������ʽ����ʱ��Һ��pH���±���
| ������ | Fe3+ | Fe2+ | Mg2+ | Al3+ | Cu2+ | Cr3+ |
| ��ʼ����ʱ��pH | 1.9 | 7.0 | ���� | ���� | 4.7 | ���� |
| ������ȫʱ��pH | 3.2 | 9.0 | 11.1 | 8 | 6.7 | 9 ����9�ܽ⣩ |
��1��ʵ������18.4 mol��L��1��Ũ��������250 mL 4.8 mol��L��1��������Һ�����õIJ����������ձ����������������ܣ�һ���ܾ�ȷ��ȡһ�����Һ����������⣬���� ��
��2�����ʱ��Ϊ����߽�ȡ�ʿɲ�ȡ�Ĵ�ʩ�� �����ٴ�һ�㣩��
��3��������Һ��pH=8��Ϊ�˳�ȥ ���ӡ������ӽ�����֬��ԭ��Ϊ��
Mn����nNaR��MRn��nNa�����������ĵ����������� ��
��4��Cr(OH)3����Һ�д������³����ܽ�ƽ�⣺Cr(OH)3(s)
 Cr3+(aq)+3OH��(aq)
Cr3+(aq)+3OH��(aq)�����£�Cr(OH)
 ���ܶȻ�Ksp= c(Cr3+)? c3(OH��)=10��32��Ҫʹc(Cr3+)����10��5mol/L����Һ��pHӦ���� ��
���ܶȻ�Ksp= c(Cr3+)? c3(OH��)=10��32��Ҫʹc(Cr3+)����10��5mol/L����Һ��pHӦ���� ����5����ԭ���̷������·�Ӧ������ƽ���� Na2Cr2O7�� SO2��
= Cr(OH) (H2O)5SO4�� Na2SO4��
��ҵ��ˮ�г�����һ������Cr2O72����CrO42�������ǻ�����༰��̬ϵͳ�����ܴ���������д�����
����һ�ִ�������Ϊ��ⷨ���÷���Fe���缫����⺬Cr2O72�������Է�ˮ�����ŵ����У�����������ҺpH���ߣ�����Cr(OH)3��������Fe���缫��ԭ��Ϊ
������������ҺpH���ߵ�ԭ���ǣ��õ缫��Ӧ���ͣ� ��