��Ŀ����
����Ŀ��A��B��CΪ������Ԫ�أ������ڱ���������λ����ͼ��ʾ��A��C��Ԫ�ص�ԭ�Ӻ��������֮�͵���Bԭ�ӵ���������Bԭ�Ӻ�������������������ȡ�
![]()
(1)д��A��B��C��Ԫ������________��________��________��
(2)C��Ԫ�����ڱ��е�λ����____________________��
(3)B��ԭ�ӽṹʾ��ͼΪ________________��C���⻯����B���⻯����ȶ���ǿ��˳��Ϊ________>________(�ѧʽ)��
(4)�Ƚ�A��C��ԭ�Ӱ뾶A________C��д��A����̬�⻯����A������������Ӧˮ���ﷴӦ�Ļ�ѧ����ʽ______________________��
���𰸡���1���� �� ��
��2���ڶ�������A��
��3��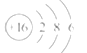 HF H2S
HF H2S
��4��NH3+HNO3=NH4NO3
��������
��A��B��C�����ڱ��е�λ�ÿ�֪��A��C���ڵڶ����ڣ�B���ڵ������ڣ���B��ԭ������Ϊx����AΪx-9��CΪx-7����������x-9+x-7=x����x=16��������Bԭ�Ӻ�������������������ȣ���B��������Ϊ16����S����ôAΪN��CΪF��

��ϰ��ϵ�д�
 �ľ�ͼ���ʱ�ȷ�ϵ�д�
�ľ�ͼ���ʱ�ȷ�ϵ�д�
�����Ŀ