��Ŀ����
����ʥ·��˹��ѧ�������͵��Ҵ�ȼ�ϵ�أ�������(H+)�ܼ�����200oC���ҹ��硣����ܷ�ӦΪ��C2H5OH+3O2��2CO2+3H2O����ͼ�Ǹõ�ص�ʾ��ͼ������˵����ȷ����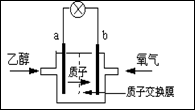
| A��a��Ϊ��ص����� |
| B����ع���ʱ������a���ص��߾������ٵ�b�� |
| C����������ĵ缫��ӦΪ��4H+ + O2 + 4e��=2H2O |
| D����ع���ʱ��1mol�Ҵ�������ʱ����6mol����ת�� |
C
�������������A��ԭ��ع���ʱ���������������ƶ�����aΪ����������B����ع���ʱ�����������������·�������ڸõ������b������a��������C�����������õ����ӱ���ԭ���缫��ӦʽΪ4H++O2+4e-=2H2O����ȷ��D���Ҵ���CԪ�صĻ��ϼ�Ϊ-2�ۣ������������ߵ�+4�ۣ����ع���ʱ��1mol�Ҵ�������ʱ����12mol����ת�ƣ�����
���㣺���⿼��ԭ���ԭ����

ij��ѧʵ��С����г�������һƿijƷ��ʳ�ð״ף���Ҫ�Ǵ����ˮ��Һ������ʵ���ұ�NaOH��Һ������еζ��Բⶨ����ȷŨ�ȡ��±���4�ֳ���ָʾ���ı�ɫ��Χ��
| ָʾ�� | ʯ�� | ���� | ���� | ��̪ |
| ��ɫ��Χ��pH�� | 5��0��8��0 | 3��1��4��4 | 4��4��6��2 | 8��2��10��0 |
��1����ʵ��Ӧѡ�� ��ָʾ����
��2����ͼ��ʾ50mL�ζ�����Һ���λ�ã���A��C�̶ȼ����1mL��A���Ŀ̶�Ϊ25���ζ�����Һ�����ӦΪ mL����Ӧ��ζ��յ�ʱ������Ϊ ��
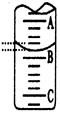
��3��Ϊ��Сʵ������ͬѧһ������������ʵ�飬����ÿ����ȡ�״������ΪVmL��NaOH��ҺŨ��Ϊc mo1/L������ʵ������¼���£�
| ʵ����� | ��һ�� | �ڶ��� | ������ |
| ����NaOH��Һ���/mL | 26��02 | 25��35 | 25��30 |
���ϱ����Կ�������һ��ʵ���м�¼����NaOH��Һ��������Զ��ں����Σ���ԭ������� ��
A��ʵ�����ʱ�����ӿ̶��߶�ȡ�ζ��յ�ʱNaOH��Һ�������
B���ζ�ǰ�ζ��ܼ��������ݣ��ζ��������첿�ֳ�����Һ��
C��ʢװ�״���Һ�ĵζ���������ˮϴ����δ�ð״���Һ��ϴ��
D����ƿԤ����ʳ�ð״���ϴ����
E���μ�NaOH��Һʱ��δ������տ�����Һ��ɫ������ֹͣ�ζ���
��4�������������ݣ�д������ð״��д�������ʵ���Ũ�ȵı���ʽ(���û���)��C1�� mol/L��
ijѧ����0.1mol/LNaHCO3��Һ�еμӷ�̪��Һ1�Σ�������Һ����û��ʲô�仯������Һ���Ⱥ������Ե���ɫ�����Ƚϳ�ʱ�����ȴ����ɫ����ȥ��
��ѧ��Ϊ���˽�ù��̵�ԭ����������̽�����̣�
��ʵ��̽����
ʵ��1: ����0.1mol/LNaHCO3��Һ�������ҺpH�仯���±�
| �¶ȣ��棩 | 10 | 20 | 30 | 50 | 70 | 80 | 100 |
| pH | 8.3 | 8.4 | 8.5 | 8.9 | 9.4 | 9.6 | 10.1 |
ʵ��2��10��ʱ�����ձ��м���0.1mol/LNaHCO3��Һ200mL����ø���ҺpH=8.3�����ȵ�100�棬���pH=10.2���ָ���10�棬pH=9.8��
ʵ��3������0.1mol/LNaHCO3��Һ��������������ͨ�����ʯ��ˮ����Һ����ǡ�
ʵ��4��
������0.1mol/LNaHCO3��Һ��0.1mol/LNa2CO3��Һ��200mL��10��ʱ���ֱ���NaHCO3��ҺpH=8.3��Na2CO3��ҺpH=11.5��
�ڼ�������0.1mol/L NaHCO3��Һ200mL������Һ���100mL��ֹͣ���ȣ���ˮ��200mL����ȴ��ԭ�¶ȣ������ҺpH=9.8��
�۽�0.1mol/L NaHCO3��Һ200mL���ڷ������죬�ټ�ˮ��200mL�������ҺpH=10.1��
���������ʵ��ش��������⣺
��1�������ӷ���ʽ��ʾ0.1mol/LNaHCO3��Һ�д��ڵ�ƽ�⣨��ˮ����ƽ���⣩___________
________________��_______________________________��������ƽ����_______________
����������_______________________________.
��1��ʵ��3�õ��Ľ�����__________________________________________________��
��1�����ʵ��2��3����������0.1mol/LNaHCO3��Һ��pH�����ԭ�������_____________
__________��______________________��________________________________��
��1��ʵ��4�ٵõ��Ľ�����_______________________________________________��
��1��ʵ��4�ڡ���ˮ��200mL����Ŀ����____________________________________________��
ʵ��4�ۿ��Եõ��Ľ�����_______________________________________��
��1��Ҫȷ��NaHCO3��Һ���Ⱥ�pH�������Ҫԭ����Ҫ�����������________________��
��ͼ��ʾװ����,a?b���Ƕ��Ե缫,ͨ��һ��ʱ���(���Һ����),a��������Һ�Ժ�ɫ?����˵����ȷ����
| A��a�Ǹ���,b������ |
| B��CuSO4��Һ��pH��С |
C��ͭ�缫�ϵķ�ӦʽΪ |
| D����NaCl��Һ�м���һ����������,��ʹ��Һ��ԭ����Һ��ȫһ�� |
������������Դ���������й㷺��;��������Ni���������缫���ŨNaOH��Һ�Ʊ���������Na2FeO4��װ����ͼ��ʾ�������ƶϺ������ǣ� ����
| A�������������缫��ӦΪFe��6e-+4H2O=FeO42-+ 8H+ |
| B�����ʱ���ӵ���������Ϊ��������Ni�缫����Һ��Fe�缫������ |
| C������ĤΪ�����ӽ���Ĥ����OH-���������ƶ� |
| D�����ʱ������pH���͡�������pH���ߣ���ȥ��Ĥ��Ϻ���ԭ��Һ�Ƚ�pH���ͣ�������ǰ������仯���Բ��ƣ� |
������ͼ��ʾ��ԭ��أ�����˵����ȷ���� 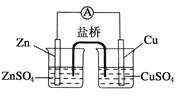
| A��п�缫������ԭ��Ӧ��ͭ�缫����������Ӧ |
| B�������е�������������ͭ��Һ��Ǩ�� |
| C�����Ӵ�п�缫ͨ������������ͭ�缫 |
| D��ͭ�缫�Ϸ����ĵ缫��ӦΪ2H����2e��===H2�� |
������ʵ�����õ绯ѧԭ�����͵��ǣ� ��
| A����п��Ƭ�ȶ�����Ƭ����ʴ |
| B��Զ���ֵ�β��װ��һ��������п�� |
| C�����������£��ڿ����������ױ���ʴ |
| D����п������������ʱ���������е���������ͭ |
ȼ�ϵ����ȼ�ϣ���H2��CO��CH4�ȣ��������������Ӧ������ѧ��ת��Ϊ���ܵ�װ�ã����ҺΪǿ����Һ�����й��ڼ���ȼ�ϵ�ص�˵����ȷ���ǣ� ��
| A��������ӦΪCH4+10OH����CO32��+7H2O+8e�� |
| B��������ӦΪO2+2H2O+4e����4OH�� |
| C�����ŷŵ���У���Һ�е����������ƶ� |
| D�����ŷŵ���У���Һ��PH���� |
�����йص�ص�˵������ȷ����
| A���ֻ����õ�����ӵ�����ڶ��ε�� |
| B��ͭпԭ��ع���ʱ�����������·��ͭ�缫����п�缫 |
| C���״�ȼ�ϵ�ؿ�ֱ�Ӱѻ�ѧ��ת��Ϊ���� |
| D��Ǧ���س��ʱ�������ķ�ӦΪ��PbSO4��2H2O��2e-��PbO2��SO42-��4H+ |