��Ŀ����
BaCl2?xH2O�нᾧˮ��Ŀ��ͨ����������ȷ����
�ٳ�ȡ1.222g��Ʒ������С�ձ��У���������ϡ���ᣬ�����ܽ⣬�߽���ߵμ�ϡ���ᵽ������ȫ�����ã�
�ڹ��˲�ϴ�ӳ�����
�۽�ʢ�г�������ֽ����ɲ��������գ�ת�����¯�У��������յ����أ��Ƶó�������Ϊ1.165g��
�ش��������⣺
��1���ڲ������У���Ҫ�Ⱥ���ϡ�����ϴ�ӳ���������������������Ƿ�ϴ���ķ����� ��
��2������BaCl2?xH2O�е�x= ����Ҫ��д��������̣���
��3���������У����������������¶ȹ��ߣ����ܻ��в��ֳ�������ֽ�е�̼��ԭΪBaS����ʹx�IJⶨ��� ���ƫ�͡�����ƫ�ߡ����䡱����
�ٳ�ȡ1.222g��Ʒ������С�ձ��У���������ϡ���ᣬ�����ܽ⣬�߽���ߵμ�ϡ���ᵽ������ȫ�����ã�
�ڹ��˲�ϴ�ӳ�����
�۽�ʢ�г�������ֽ����ɲ��������գ�ת�����¯�У��������յ����أ��Ƶó�������Ϊ1.165g��
�ش��������⣺
��1���ڲ������У���Ҫ�Ⱥ���ϡ�����ϴ�ӳ���������������������Ƿ�ϴ���ķ�����
��2������BaCl2?xH2O�е�x=
��3���������У����������������¶ȹ��ߣ����ܻ��в��ֳ�������ֽ�е�̼��ԭΪBaS����ʹx�IJⶨ���
���㣺��ѧ����ʽ���йؼ���
ר�⣺������,ʵ�������
��������1��Ӧ������������ˮ���н��г���ϴ�ӣ����巽����ȡˮϴҺ���Թ��У�����ϡ�����ữ���μ�AgNO3��Һ�����ް�ɫ���dz��֣������Cl-�Ѿ�������
��2���������ᱵ���������Ȼ�����������������ˮ�����������ݶ�������֮��ȷ��x��ֵ��
��3�����ֳ�������ֽ�е�̼��ԭΪBaS�����ʹ������ᱵ������ƫС���ⶨ�Ȼ���������ƫС��ˮ������ƫ��
��2���������ᱵ���������Ȼ�����������������ˮ�����������ݶ�������֮��ȷ��x��ֵ��
��3�����ֳ�������ֽ�е�̼��ԭΪBaS�����ʹ������ᱵ������ƫС���ⶨ�Ȼ���������ƫС��ˮ������ƫ��
���
�⣺��1��Ӧ������������ˮ���н��г���ϴ�ӣ����巽����ȡˮϴҺ���Թ��У�����ϡ�����ữ���μ�AgNO3��Һ�����ް�ɫ���dz��֣������Cl-�Ѿ�������
���ʴ�Ϊ������ˮ��ȡˮϴҺ���Թ��У�����ϡ�����ữ���μ�AgNO3��Һ�����ް�ɫ���dz��֣������Cl-�Ѿ�������
��2������1.165gΪ���ᱵ�����������ʵ���=
=0.005mol�����Ȼ����������Ȼ���������=0.005mol��208g/mol=1.04g�����нᾧˮ������=1.222g-1.04g=0.182g����208��18x=1.04g��0.182g�����x��2���ʴ�Ϊ��2��
��3�����ֳ�������ֽ�е�̼��ԭΪBaS�����ʹ������ᱵ������ƫС���ⶨ�Ȼ���������ƫС��ˮ������ƫ��x��ֵƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
���ʴ�Ϊ������ˮ��ȡˮϴҺ���Թ��У�����ϡ�����ữ���μ�AgNO3��Һ�����ް�ɫ���dz��֣������Cl-�Ѿ�������
��2������1.165gΪ���ᱵ�����������ʵ���=
| 1.165g |
| 233g/mol |
��3�����ֳ�������ֽ�е�̼��ԭΪBaS�����ʹ������ᱵ������ƫС���ⶨ�Ȼ���������ƫС��ˮ������ƫ��x��ֵƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
���������⿼��������ɵIJⶨ������ʽ��ȷ�����ѵ㣬��Ҫѧ���߱���ʵ�Ļ������Ѷ��еȣ�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
�Ȼ�ԭ��ұ�������ķ�Ӧһ���ǣ�������
| A��������ԭ��Ӧ | B���û���Ӧ |
| C���ֽⷴӦ | D�����ֽⷴӦ |
����ĸ����ǣ�������
| A������̼���⡢������Ԫ�ص��л��� |
| B������ͨʽCn��H2O��m�Ļ����� |
| C����������� |
| D��һ���Ƕ��ǻ�ȩ�����ǻ�ͪ�Լ���ˮ���������ǵ����� |


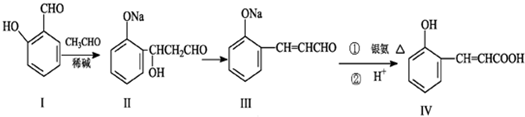
 ��
��