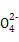��Ŀ����
�������ӷ���ʽ�У���ȷ���ǣ� ��
| A����Na2O2����Ͷ��H218O�У�2H218O��2Na2O2 = 4OH����4Na����18O2�� |
| B��1mol��L��NaAlO2��Һ��2.5 mol��L��HCl�����������Ȼ�ϣ�2AlO2�� + 5 H��=Al(OH)3����Al3����H2O |
| C��̼��������Һ����������������Һ��ϣ�HCO3��+Ba2��+OH��=BaCO3��+H2O |
| D����������Һ�������Һ��ϣ�SiO32��+ 2H�� = H2SiO3�� |
B
���������A������Na2O2������������ʱ��ԭ����ˮû�в���������ԭ��Ӧ��18OӦ�������������У�B����ȷ��C����������������Ӧ����ȫ��Ӧ��Ba2����OH���Ĺ�ϵӦΪ1:2��D�����������ᣬ���ܲ�

��ϰ��ϵ�д�
�����Ŀ
 Cl2����H2����2OH��
Cl2����H2����2OH�� KI3
KI3


 Na++H++C
Na++H++C
 4OH-
4OH-