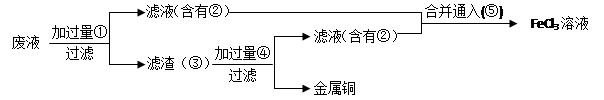
��Ŀ����
��ҵ��������������������Ҫ�ɷ�ΪFe2O3��FeO��SiO2�ȣ�Ϊԭ���Ʊ��ߵ���������(Fe2O3 )�����������������£�
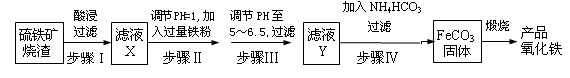
�Իش��������⣺
��1����ҺX�к��еĽ����������� �������ӷ��ţ���
��2��������п�ѡ�� �Լ�������Һ��pH������ĸ����
��3�����˲����г��˲��������ձ�����Ҫ�IJ��������� ��
��4��������ķ�Ӧ�¶�һ���������35�����£���Ŀ���� ��
��5���ڿ���������FeCO3���ɲ�Ʒ�������Ļ�ѧ����ʽΪ ��

�Իش��������⣺
��1����ҺX�к��еĽ����������� �������ӷ��ţ���
��2��������п�ѡ�� �Լ�������Һ��pH������ĸ����
| A��ϡ���� | B����ˮ | C������������Һ | D�����������Һ |
��4��������ķ�Ӧ�¶�һ���������35�����£���Ŀ���� ��
��5���ڿ���������FeCO3���ɲ�Ʒ�������Ļ�ѧ����ʽΪ ��
��1��Fe2+��Fe3+��2��B
��3����ͨ©��
��4����ֹNH4HCO3�ֽ⣬����Fe2+��ˮ��
��5��4FeCO3 + O2 2Fe2O3 + 4CO2
2Fe2O3 + 4CO2
��3����ͨ©��
��4����ֹNH4HCO3�ֽ⣬����Fe2+��ˮ��
��5��4FeCO3 + O2
 2Fe2O3 + 4CO2
2Fe2O3 + 4CO2�����������1������Fe2O3��FeO��Ӧ������Fe2+��Fe3+������SiO2����Ӧ��
��2������PHֵ����Ҫ�����������ƻ��Fe2+���ӷ�����Ӧ���ɳ�������ѡB��
��3�����˲����õ��IJ�������Ϊ����ͨ©�������������ձ���
��4���¶Ȳ�̫�ߣ�Ϊ���Ƿ�ֹNH4HCO3�ֽ⣬����Fe2+��ˮ�⡣
��5�������ṩ�ķ�Ӧ����������д�ɻ�ѧ����ʽ��
�����������ʱҪ���ջ�ѧ����ʽ����д������ʵ�鷽���֪ʶ��ֻ���������ܶ�����������ȷ���жϡ�

��ϰ��ϵ�д�
�����Ŀ