��Ŀ����
ij���Ժ� Na+����Һ�У����ܻ�����NH4+��Fe2+��Br-��CO32-��I-��SO32-���������е�һ�ֻ��֡���������ʵ�飺����ԭ��Һ�еμ�������ˮ�������ݲ�������Һ�ʳȻ�ɫ������Ȼ�ɫ��Һ�м���BaCl2��Һʱ���������ɣ��۳Ȼ�ɫ��Һ����ʹ���۱������ɴ˿�����֪����Һ�п϶������ڵ�������
| A��NH4+��Br-��CO32- | B��NH4+��I-��SO32- |
| C��Fe2+��I-��SO32- | D��Fe2+��CO32-��I- |
A
��������������ٵμ�������ˮ�������ݲ�������Һ�ʳȻ�ɫ��˵����CO32-����û��Fe2+������Ȼ�ɫ��Һ�м���BaCl2��Һʱ���������ɣ�һ��û��SO32-���۳Ȼ�ɫ��Һ����ʹ���۱�����һ��û��I-���ɴ˿�����֪����Һ�п϶������ڵ������ǣ�Fe2+��I-��SO32-��C����ȷ��
���㣺�������ӹ���֪ʶ��

��ϰ��ϵ�д�
�����Ŀ
ij��ɫ��Һ�е���ʯ����Һ�Ժ�ɫ������Һ�п��Դ����������
| A��Na+��Cl����Mg2+��ClO- | B��Fe2+��NH4+��Cl����NO3һ |
| C��Na+��HCO3����AlO2һ��K+ | D��Na+��Ba2+��Cl����NO3һ |
���и�������һ���ܴ����������
A��ij��ɫ����������Һ��Cl����Na+��MnO4-��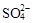 |
B����ʹpH��ֽ������ɫ����Һ��Na+��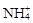 ��K+�� ��K+��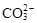 |
| C���������NaOH��Һ��ɵõ��������Һ��K+��Ba2+��HCO3-��Cl�� |
| D����ˮ���������c(H+)=10?12mol/L����Һ��K+��Ba2+��Cl����NO3�� |
��ָ�������£����и������ӿ��ܴ����������
A����ɫ��������Һ�У�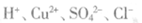 |
B��������Ӧ�ų�H2����Һ�У� |
C���μӷ�̪�Ժ�ɫ����Һ�У�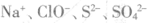 |
D����ˮ�����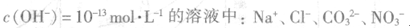 |
�������ӷ���ʽ��ȷ����
| A�������ȥˮ���е�CaCO3��CaCO3��2H+=Ca2+��H2O��CO2�� |
| B����ˮ��ͨ��������SO2��I2 +SO2+2H2O=2I��+SO42��+4H+ |
| C��NaHSO4��Һ��Ba(OH)2��Һ��Ӧ�����ԣ�H+��SO42����Ba2+��OH��=BaSO4����H2O |
| D����ǿ����Һ�д���������Fe(OH)3��Ӧ����Na2FeO4�� |
�������ӷ���ʽ��ȷ����
| A��������������Һ��ȥ��Ƭ�����ϵ�����Ĥ��Al3����4OH��=AlO2����2H2O |
| B���ں�3��2amolHNO3��ϡ��Һ�У�����amol���ۣ�5Fe��4NO3����16H��=3Fe2����2Fe3����4NO����8H2O |
| C������ʯ��ˮ�������С�մ���Һ��Ӧ��Ca2����OH����HCO3��=CaCO3����H2O |
| D������������ʹ����KMnO4��Һ��ɫ��3H2O2+2MnO4-+6H+=2Mn2++4O2��+6H2O |
�������ӷ���ʽ��д��ȷ����
| A�����Ȼ��ϡ��Һ�м���ϡ�ռ���Һ NH4+ + OH��=NH3��+ H2O |
| B����ˮ�����ᷴӦ H+ + OH��=H2O |
| C���ƺ�ˮ��ӦNa+ 2H2O= Na+ + 2OH��+ H2�� |
| D���������ռ���Һ��ӦCl2+ 2OH��=Cl��+ ClO��+ H2O |
�������ӷ���ʽ��ȷ����
| A��������þ��ϡ���ᷴӦ��H+ + OH��=H2O |
| B��AlCl3��Һ�м���������ˮ��Al3����3OH��=Al(OH)3�� |
| C��ͭ����ϡ���3Cu+8H++2NO3��=3Cu2++2NO��+4H2O |
| D�����������Һ��ͨ�����CO2��Ca2++2ClO��+H2O+CO2=CaCO3��+2HClO |