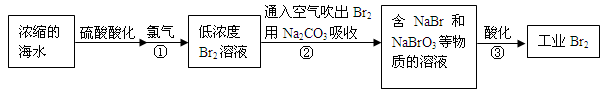
��Ŀ����
�����ڹ�ũҵ����������ҪӦ�á�
��1���ٵ������ڹ�ҵ�ϳɰ���д�������ĵ���ʽ ��
��NH3���ȶ��Ա�PH3 ����д��ǿ������������
��2������ͼ��ʾ����NaOH�����ϵμ���Ũ��ˮ��Ѹ�ٸ��ϸǣ��۲�����
��Ũ����Һ�θ�������ְ��̣�������Ӧ�Ļ�ѧ����ʽΪ ��
��Ũ����Һ���Ϸ�û����������һ��ʱ���Ũ�����Һ�����а�ɫ���壬�ù�������� ��д��ѧʽ��һ�ּ��ɣ���
��FeSO4Һ�����ȳ��ֻ���ɫ��������һ��ʱ����ɺ��ɫ�������ķ�Ӧ����
Fe2++2NH3��H2O=Fe(OH)2��+ 2NH4+ �� ��
��3���������ѷ���Ŀǰ����NH3��ˮ����Ⱦ����Ҫ��������һ�������£���ˮ���м�������NaOH��ʹNH3���ѳ���������ƽ���ƶ�ԭ��������ԭ�� ��
��4�������������£���������ˮ�зֽ�����İ��ܹ��������������������ᣨHNO2������Ӧ�Ļ�ѧ����ʽΪ ������Ӧ����0.3 mol���ӷ���ת��ʱ�����������������Ϊ g��С���������λ��Ч���֣���
��1����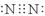 ��1�֣� ��ǿ��1�֣�
��1�֣� ��ǿ��1�֣�
��2����NH3+HCl=NH4Cl��1�֣� ��NH4HSO4��(NH4)2SO4��1�֣�
��4Fe(OH)2+O2+2H2O=4Fe(OH)3��2�֣�
��3������ˮ�д���ƽ�⣺NH3+H2O NH3��H2O
NH3��H2O NH4+ +OH-��1�֣�д��NH3+H2O
NH4+ +OH-��1�֣�д��NH3+H2O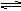 NH4+ +OH-���۷֣�������NaOH��OH-Ũ������ƽ�������ƶ���1�֣����������ڰ����ѳ���2�֣�
NH4+ +OH-���۷֣�������NaOH��OH-Ũ������ƽ�������ƶ���1�֣����������ڰ����ѳ���2�֣�
��4��2NH3+3O2 =2HNO2+2H2O��2�֣���д���ﲻ�۷֣� 2.35 g��2�֣�
=2HNO2+2H2O��2�֣���д���ﲻ�۷֣� 2.35 g��2�֣�
���������������1���ٵ���������Nԭ�Ӽ��γ�3�Թ��õ��ӣ�������Ϊ��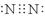
��NԪ�طǽ�����ǿ��PԪ�أ�����NH3���ȶ��Ա�PH3ǿ��
��2����Ũ����ӷ�����HCl��Ũ��ˮ�ӷ�����NH3��Ӧ�����ɵİ���ΪNH4Cl����ѧ����ʽΪ��NH3+HCl=NH4Cl��
��Ũ��ˮ�ӷ�����NH3������Ũ���������ᷴӦ���ɣ�NH4HSO4��(NH4)2SO4��
�����ձ�Ϊ���ɫ��������Fe(OH)3����ѧ����ʽΪ��4Fe(OH)2+O2+2H2O=4Fe(OH)3��
��3��NH3?H2OΪ������ڵ���ƽ�⣺NH3+H2O  NH3?H2O
NH3?H2O NH4++OH?������NaOH��������OH?Ũ�ȣ�ʹƽ�����淴Ӧ�����ƶ��������ڰ����ѳ���
NH4++OH?������NaOH��������OH?Ũ�ȣ�ʹƽ�����淴Ӧ�����ƶ��������ڰ����ѳ���
��4��NH3��O2��Ӧ�����������ˮ����ƽ�ɵû�ѧ����ʽ��2NH3+3O2 2HNO2+2H2O�����ݻ��ϼ۵ı仯��֪��6��n��HNO2��=0.3mol��n��HNO2��=0.05mol����m��HNO2��=2.35g��
2HNO2+2H2O�����ݻ��ϼ۵ı仯��֪��6��n��HNO2��=0.3mol��n��HNO2��=0.05mol����m��HNO2��=2.35g��
���㣺���⿼��������ͷ���ʽ����д��Ԫ�������ɡ�����ƽ�⡣

��ͼ1�ǹ�ҵ�ϡ���������� ʾ��ͼ����ͼ2�ǹ�ҵ�ϡ�ʯ�ҡ�ʯ����������ʾ��ͼ���ش�

��֪��
| �� �� | Ca(OH)2 | NaOH |
| �۸�Ԫ/kg�� | 0.36 | 2.9 |
| ����SO2�ijɱ���Ԫ/mol�� | 0.027 | 0.232 |
��1��ͼ1�У���Ӧ�ٵ����ӷ���ʽΪ ��֮һ�� ��
��2��ͼ2�У���Ӧ�ݵ�Ŀ�����ȶ��̻���Ԫ���γɸ�̬�Ļ���������������� ���÷����ŵ�֮һ�� ��
��3��������������еIJ���Na2SO3���ȶ��Բ��ã���ʹ��Ԫ������ת��Ϊ�ӷ�����Ⱦ�Ϊ����֮������ˢ۵ķ�Ӧ��д���仯ѧ����ʽΪ ��
����ǿ������ʾ���Ũ���йء��ش��������⣺
��1��ijѧϰС����15 mol/LŨ��������100 mL3 mol/Lϡ���ᡣ
��������Ҫ����Ͳ��ȡ15 mol/LŨ����_______mL��
������ͼ��ʾ�����������ƹ����в����õ���_________������ţ���
��ͼ�����е������⣬����������Һ�����õ��IJ���������___________��
������ʵ������У�����ȷ����_________����д��ţ���
| A��ʹ������ƿǰ��������Ƿ�©ˮ�� |
| B������ʱҺ�泬���̶��ߣ������Һ��Ӧ�ý�ͷ�ι������� |
| C��������Һʱ������Ͳ��ȡŨ����ֱ�ӵ�������ƿ�У�Ȼ�������ˮ���ݡ� |
| D�����ݺ�Ǻ�ƿ����������ƿ�������µߵ���ҡ�ȡ� |
��2����ͭ��Ũ�����ڼ��������·�Ӧ��Ũ������ֳ�����������___________��
��MnO2��Ũ�����ڼ��������·�Ӧ�����ӷ���ʽΪ__________��
�۷ֱ���Ũ�����ϡ������ȡ��ͬ����Cu(NO3)2���壬����Ũ�����ϡ��������ʵ���֮����__________��