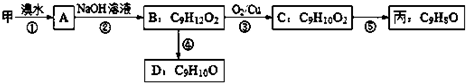
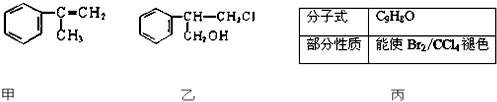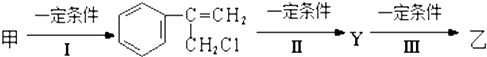��Ŀ����
 ��һ���¶��£����Ϊ2L���ܱ������У�NO2��N2O4֮�䷢����Ӧ��
��һ���¶��£����Ϊ2L���ܱ������У�NO2��N2O4֮�䷢����Ӧ��2NO2��g��������ɫ��?N2O4��g������ɫ������ͼ��ʾ��
��1������
��2���������¶ȣ���v������
��3����������Ӧ�ڼס���������ͬ������ͬʱ���У��ֱ��ü���v��NO2��=18mol/��L?min��������v��N2O4��=0.2mol/��L?s������
��4����0��1min������X��ʾ�÷�Ӧ��������
���㣺���ʵ�����Ũ����ʱ��ı仯����,��ѧƽ��ļ���
ר�⣺��ѧƽ��ר��
��������1���������ʵ����ı仯��֮�ȵ��ڼ�����֮�ȷ�����
��2�������¶ȶԷ�Ӧ���ʵ�Ӱ�������
��3�����ݷ�Ӧ����֮�ȵ��ڻ�ѧ������֮�ȱȽϣ�
��4������v=
���㷴Ӧ���ʣ����ͼ����������������
��2�������¶ȶԷ�Ӧ���ʵ�Ӱ�������
��3�����ݷ�Ӧ����֮�ȵ��ڻ�ѧ������֮�ȱȽϣ�
��4������v=
| ��c |
| ��t |
���
�⣺��1���ɷ���ʽN2O4��g��?2NO2 ��g������֪��Ӧ��NO2 �����ʵ����仯�ϴ���Y��ʾNO2�����ʵ�����ʱ��ı仯���ߣ��ʴ�Ϊ��Y��
��2���¶����ߣ����淴Ӧ���ʶ����ʴ�Ϊ���ӿ죻�ӿ죻
��3������v��NO2��=18mol/��L?min��������v ��N2O4��=0.2mol?L-1?min-1��v��NO2��=2v��N2O4��=0.4mol?L-1?s-1=24mol/��L?min�������ҷ�Ӧ�Ͽ죬
�ʴ�Ϊ���ң�
��4��v��X��=
=0.15mol?��L?min��-1��
�÷�Ӧ�������ʱ��Y��ת����Ϊ��
��100%=60%��
��ʼʱ��n��X��+n��Y��=0.4mol+1mol=1.4mol��
ƽ��ʱ��n��X��+n��Y��=0.7mol+0.4mol=1.1mol��
����ͬ�����£������ѹǿ֮�ȵ������ʵ���֮�ȣ�
��Ӧ��ʼʱ�뷴Ӧ��ƽ��״̬ʱ��ѹǿ֮��Ϊ1.4mol��1.1mol=14��11��
�ʴ�Ϊ��0.1mol?��L?min��-1��60%��14��11��
��2���¶����ߣ����淴Ӧ���ʶ����ʴ�Ϊ���ӿ죻�ӿ죻
��3������v��NO2��=18mol/��L?min��������v ��N2O4��=0.2mol?L-1?min-1��v��NO2��=2v��N2O4��=0.4mol?L-1?s-1=24mol/��L?min�������ҷ�Ӧ�Ͽ죬
�ʴ�Ϊ���ң�
��4��v��X��=
| ||
| 1min |
�÷�Ӧ�������ʱ��Y��ת����Ϊ��
| 1mol-0.4mol |
| 1mol |
��ʼʱ��n��X��+n��Y��=0.4mol+1mol=1.4mol��
ƽ��ʱ��n��X��+n��Y��=0.7mol+0.4mol=1.1mol��
����ͬ�����£������ѹǿ֮�ȵ������ʵ���֮�ȣ�
��Ӧ��ʼʱ�뷴Ӧ��ƽ��״̬ʱ��ѹǿ֮��Ϊ1.4mol��1.1mol=14��11��
�ʴ�Ϊ��0.1mol?��L?min��-1��60%��14��11��
���������⿼���Ϊ�ۺϣ���Ŀ�Ѷ��еȣ�ע����ջ�ѧ��Ӧ�����Լ���ѧƽ��Ļ���֪ʶ���ɽ����⣮

��ϰ��ϵ�д�
�����Ŀ
���������ķ�0.1mol?L-1�Ĵ�������Һ�зֱ�������������ᡢ�������ƹ��塢�����ƹ��塢�������ƹ��壨������Һ������¶ȱ仯��������Һ��
�仯��ȷ���ǣ�������
| c(Ac-) |
| c(Na+) |
| A�����������䡢��С |
| B������С������С |
| C����С����������С |
| D������С�����䡢��С |
����ѡ���йط�����������ȷ���ǣ�������
| A��������֬������������ˮ����Ƶ� |
| B����������Ҫ�ɷ��Ǹ�֬�����ƺ��� |
| C��ʹ��������֬ˮ���Ļ�����з���Ĺ��������� |
| D�������Ƴɺ�������ˮ�γɽ��� |
 �����£���pH=2�Ĵ�����Һ��pH=2�������ң���������в����ش��й����⣺
�����£���pH=2�Ĵ�����Һ��pH=2�������ң���������в����ش��й����⣺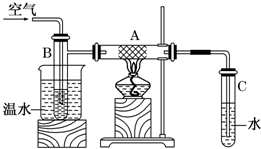 ��ͼ��Ӳ���Թ�A�з������ྻ��ϸͭ˿���ձ��з�����ˮ���Թ�B�з���״����ҷ��Թ�C�з�����ˮ����B�в��Ϲ��������ʹ�״������Ϳ���ͨ�����ȵ����ȳ̶ȵ�ͭ˿��
��ͼ��Ӳ���Թ�A�з������ྻ��ϸͭ˿���ձ��з�����ˮ���Թ�B�з���״����ҷ��Թ�C�з�����ˮ����B�в��Ϲ��������ʹ�״������Ϳ���ͨ�����ȵ����ȳ̶ȵ�ͭ˿��