��Ŀ����
7��ʵ�����Ʊ�����ͪ�Ļ�ѧ����ʽΪ��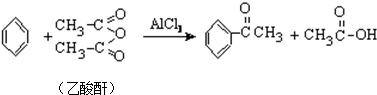
�Ʊ������л���CH3COOH+AlCl3��CH3COOAlCl2+HCl���ȸ���Ӧ��
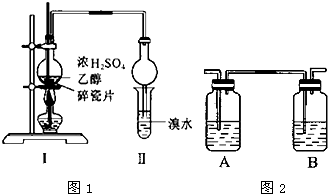
��Ҫʵ��װ�úͲ�����ͼ1��
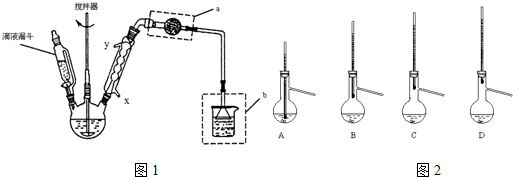
���ϳɣ�������ƿ�м��� 20g��ˮAlCl3��30mL��ˮ����Ϊ���ⷴӦҺ���¹��죬�߽���������μ�6mL��������10mL��ˮ���Ļ��Һ�����Ƶμ����ʣ�ʹ��ӦҺ�����������μ���Ϻ���Ȼ���1Сʱ��
���������ᴿ��
�ٱ߽���������μ�һ����Ũ�������ˮ���Һ������õ��л���
��ˮ���ñ���ȡ����Һ
�۽��٢������л���ϲ���ϴ�ӡ������ȥ�����õ�����ͪ�ֲ�Ʒ
������ֲ�Ʒ�õ�����ͪ���ش��������⣺
��1�������������У�����ˮӦ��x���x������y������ͨ�룻����a������Ϊ����ֹ�ձ��е�ˮ������������ƿ�У�װ��b�����ã�����HCl���壮
��2�������������ͱ��Ļ��Һһ���Ե�������ƿ�����ܵ���AD��
A����Ӧ̫���ҡ� B��Һ��̫������� �� C����Ӧ�仺�� �� D������������
��3��������ᴿ�����ڵ�Ŀ���ǰ��ܽ���ˮ�еı���ͪ��ȡ�����Լ�����ʧ���ò������Ƿ�ɸ����Ҵ���ȡ������ǡ�����ԭ�����Ҵ���ˮ���ܣ�
��4���ֲ�Ʒ�����ᴿʱ������װ��ͼ2���¶ȼ�λ����ȷ����C�����ܻᵼ���ռ����IJ�Ʒ�л��еͷе����ʵ�װ����AB��
���� ��1������������ȴЧ�����жϽ�ˮ�ڣ�����װ��ͼ�е������������Լ��ж����������ƺ����ã�
��2���������ͱ���Ӧ���ң��¶ȹ������ɸ���ĸ����
��3��������ȡ��ԭ�������������ڻ������ܵ��ܼ��� ���ܽ�Ȳ�ͬ���Ի����Һ���з��룻
��4����������װ�õ�Ŀ���ǿ������ʵķе㣬��һ���¶���������ʣ��¶ȼ�ˮ�����Dzⶨ������ƿ֧�ܿڴ��������¶ȣ�
��� �⣺��1������������ȴЧ���ÿ�֪��ˮ��Ϊx������aΪ����ܿ�����ˮ����ֹ�ձ��е�ˮ������������ƿ�У�װ��b�����������շ�Ӧ��������������HCl���壻
�ʴ�Ϊ��x����ֹ�ձ��е�ˮ������������ƿ�У�����HCl���壻
��2�������������ͱ��Ļ��Һһ���Ե�����ƿ�������ܻᵼ�·�Ӧ̫���ң���ӦҺ���¹��쵼�¸���ĸ����
�ʴ�Ϊ��AD��
��3��ˮ���ñ���ȡ����Һ��Ŀ���ǰ��ܽ���ˮ�еı���ͪ��ȡ�����Լ�����ʧ�������Ҵ�����ˮ���ܲ��ֲ㣬���Բ����þƾ����汽������ȡ������
�ʴ�Ϊ�����ܽ���ˮ�еı���ͪ��ȡ�����Լ�����ʧ�����Ҵ���ˮ���ܣ�
��4���ֲ�Ʒ�����ᴿʱ���¶ȼƵ�ˮ����Ҫ����������ƿ֧�ܿڴ���©��װ���е��¶ȼ�λ����ȷ����C����¶ȼ�ˮ�������֧�ܿ�����λ�ã��ᵼ���ռ��IJ�Ʒ�� ���еͷе����ʣ����¶ȼ�ˮ�������֧�ܿ�����λ�ã��ᵼ���ռ��IJ�Ʒ�л��и߷е����ʣ�����A��B���װ�������µͷе����ʻ����ռ����IJ�Ʒ�У�
�ʴ�Ϊ��C��AB��
���� ���⿼���������Ʊ������롢�ᴿ����ȡ��ѡ������ʹ�õ��������֪ʶ��Ӧ�ã���ѧʵ�����֪ʶ�ͻ������ܵ������ǽ���ؼ�����Ŀ�Ѷ��еȣ�

 ʵ�����Ʊ�1��2-��������ķ�Ӧԭ�����£�
ʵ�����Ʊ�1��2-��������ķ�Ӧԭ�����£�CH3CH2OH$��_{170��}^{H_{2}SO_{4}��Ũ��}$CH2=CH2
CH2=CH2+Br2��BrCH2CH2Br
���ܴ��ڵ���Ҫ����Ӧ�У��Ҵ���Ũ����Ĵ�������l40����ˮ�������ѣ�������������������Ҵ��Ʊ�1��2-���������װ�ü��й������б����£�
| �Ҵ� | 1��2-�������� | ���� | |
| ״̬ | ��ɫҺ�� | ��ɫҺ�� | ��ɫҺ�� |
| �ܶ�/g•cm-3 | 0.79 | 2.2 | 0.71 |
| �е�/�� | 78.5 | 132 | 34.6 |
| �۵�/�� | -l30 | 9 | -1l6 |
��1���ڴ��Ƹ�ʵ���У�Ҫ������Ѹ�ٵذѷ�Ӧ�¶���ߵ�170�����ң�������ҪĿ����B������ȷѡ��ǰ����ĸ��
A��������Ӧ B�����ٸ����� C����ֹ�Ҵ��ӷ�
��2����װ��C��Ӧ����C����Ŀ�������շ�Ӧ�п������ɵ��������壨����ȷѡ��ǰ����ĸ��
A������̼��������Һ B��Ũ���� C������������Һ
��3���ж�װ��D�и��Ʊ���Ӧ�Ѿ�������������ǹ۲���ˮ��ɫ�Ƿ��ʾ�
��4����1��2-��������ֲ�Ʒ���ڷ�Һ©���м�ˮ�����ã�����Ӧ���²㣨��ϡ����¡���
��5�������������������������ѣ���ȥ���ѵķ�����������ʵ�����������
| A�� | ��ˮ�ĵ����Ժܲ����ˮ���ǵ���� | |
| B�� | SO3ˮ��Һ�ĵ����Ժܺã�����SO3�ǵ���� | |
| C�� | Һ̬��ͭ�����Ժܺã�����ͭ�ǵ���� | |
| D�� | Na2OҺ̬ʱ�ܵ��磬����Na2O�ǵ���� |
��1��ʵ������ͨ����NaOH��Һ����ϴ�����ᴿ������100mL 3mol•L-1��NaOH��Һ���ձ�״����4.48L CO2ʱ��������Һ�и�����Ũ���ɴ�С��˳��Ϊc��Na+����c��HCO3-����c��CO32-����c��OH-����c��H+����
��2�������£���һ�������0.1mol•L-1�Ĵ�����Һ�м�ˮϡ�ͺ�����˵����ȷ����CD��
A����Һ�е������ӵ���Ŀ���� B������ĵ���̶�����c��H+��������
C����Һ��
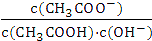 ���� D����Һ��
���� D����Һ��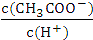 ��С
��С��3�������½�0.15mol•L-1��ϡ����V1mL��0.1mol•L-1��NaOH��ҺV2mL��ϣ�������Һ��pHΪ1����V1��V2=1��1����Һ����ı仯���Բ��ƣ���
��4�������£�Ũ�Ⱦ�Ϊ0.1mol•L-1������������Һ��pH�����ʾ��
| ���� | CH3COONa | NaHCO3 | Na2CO3 | NaClO | NaCN |
| pH | 8.8 | 9.7 | 11.6 | 10.3 | 11.1 |
A��HCNB��HClO C��H2CO3D��CH3COOH
�ڸ����������ݣ��ж����з�Ӧ���Գ�������AB��
A��CH3COOH+Na2CO3=NaHCO3+CH3COONa B��CH3COOH+NaCN=CH3COONa+HCN
C��CO2+H2O+2NaClO=Na2CO3+2HClO D��NaHCO3+HCN=NaCN+H2O+CO2��
��5���������ӿ�ʼ����ʱ��pH���±���
| ���� | Fe2+ | Cu2+ | Mg2+ |
| pH | 7.6 | 5.2 | 10.4 |
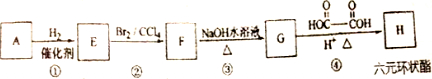
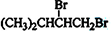 ��
�� 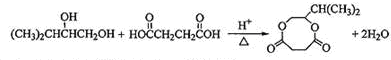 ��
�� l��2��������������Ϳ����������Ӽ���������������ɫҺ�壬�ܶ�2.189cm-3���е�131.4�棬�۵�9.79�棬������ˮ�������ڴ����ѡ���ͪ���л��ܼ�����ʵ�����п������з�Ӧ�Ʊ�1��2�������飮
l��2��������������Ϳ����������Ӽ���������������ɫҺ�壬�ܶ�2.189cm-3���е�131.4�棬�۵�9.79�棬������ˮ�������ڴ����ѡ���ͪ���л��ܼ�����ʵ�����п������з�Ӧ�Ʊ�1��2�������飮 ij�о���ѧϰС��Ϊ�ϳ�1-�������������ϵ�֪һ���ϳ�·�ߣ�
ij�о���ѧϰС��Ϊ�ϳ�1-�������������ϵ�֪һ���ϳ�·�ߣ�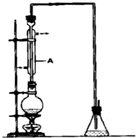 ��������һ����Ҫ���л�����ԭ�ϣ���е�Ϊ38.4�森�Ʊ��������һ�ַ������Ҵ��������ᷴӦ��ij����С������ʵ�����Ʊ��������װ������ͼ��ʵ������������£�
��������һ����Ҫ���л�����ԭ�ϣ���е�Ϊ38.4�森�Ʊ��������һ�ַ������Ҵ��������ᷴӦ��ij����С������ʵ�����Ʊ��������װ������ͼ��ʵ������������£�