��Ŀ����
����Ŀ����1������CO2��CH4�����������Ի�úϳ�������Ҫ�ɷ�ΪCO��H2�������ɼ�������������ŷţ�1����֪���������в��ַ�Ӧ���Ȼ�����ʽΪ��
��CH4��g��=C��s��+2H2��g�� ��H>0
��CO2��g��+H2��g��=CO��g��+H2O��g�� ��H>0
��CO��g��+H2��g��=C��s��+H2O��g�� ��H<0
�̶�n��CO2��=n��CH4�����ı䷴Ӧ�¶ȣ�CO2��CH4��ƽ��ת���ʼ�ͼ�ס�
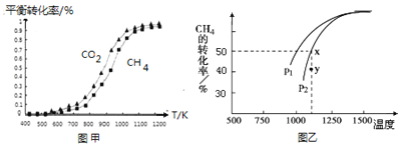
ͬ�¶���CO2��ƽ��ת����________��������������С����������������CH4��ƽ��ת���ʡ�
��2�����ܱ�������ͨ�����ʵ�����Ϊ0.1mol��CH4��CO2����һ�������·�����ӦCO2��g�� + CH4��g�� ![]() 2CO��g�� + 2H2��g����CH4��ƽ��ת�������¶ȼ�ѹǿ����λPa���Ĺ�ϵ��ͼ����ʾ��y�㣺v������_____v���棩��������������С��������������������֪�����ѹ��p����=������ѹ��p��������������ʵ�����������ƽ���ѹ����ƽ��Ũ�ȿ��Եõ�ƽ�ⳣ��Kp����x���Ӧ�¶��·�Ӧ��ƽ�ⳣ��Kp=__________________��
2CO��g�� + 2H2��g����CH4��ƽ��ת�������¶ȼ�ѹǿ����λPa���Ĺ�ϵ��ͼ����ʾ��y�㣺v������_____v���棩��������������С��������������������֪�����ѹ��p����=������ѹ��p��������������ʵ�����������ƽ���ѹ����ƽ��Ũ�ȿ��Եõ�ƽ�ⳣ��Kp����x���Ӧ�¶��·�Ӧ��ƽ�ⳣ��Kp=__________________��
���𰸡����� ���� ![]()
��������
��1������ͼʾ���Կ�����CO2��������������Ӧ��
��2���ٷ�ӦCO2��g��+CH4��g��![]() 2CO��g��+2H2��g���Ǹ������������ķ�Ӧ������ѹǿ�Ի�ѧƽ���Ӱ������
2CO��g��+2H2��g���Ǹ������������ķ�Ӧ������ѹǿ�Ի�ѧƽ���Ӱ������
��CO2��g��+CH4��g��![]() 2CO��g��+2H2��g������������ʽ����ƽ��ʱ��������ʵ���������ѹǿ֮�ȵ������ʵ���֮�ȼ���ƽ��ʱ��ѹǿ���ټ������ַ�ѹ������Kp=
2CO��g��+2H2��g������������ʽ����ƽ��ʱ��������ʵ���������ѹǿ֮�ȵ������ʵ���֮�ȼ���ƽ��ʱ��ѹǿ���ټ������ַ�ѹ������Kp= ���㣻
���㣻
��1������ͼʾ���Կ�����CO2��������������Ӧ��ͬ�¶���CO2��ƽ��ת���ʴ���CH4��ƽ��ת���ʣ��ʴ�Ϊ�����ڣ�
��2����ӦCO2��g��+CH4��g��![]() 2CO��g��+2H2��g���Ǹ������������ķ�Ӧ��ѹǿԽ��CH4��ת����ԽС����ͼ��֪��ͬ�¶��£�P1�����µ�ת���ʴ���P2����P1С��P2��ѹǿΪP2ʱ����Y�㷴Ӧδ�ﵽƽ�⣬��Ӧ�����ƶ�������v��������v���棩����ͼ֪�����¶���X����������ƽ��ʱ�����ת����Ϊ50%��������ʽ��
2CO��g��+2H2��g���Ǹ������������ķ�Ӧ��ѹǿԽ��CH4��ת����ԽС����ͼ��֪��ͬ�¶��£�P1�����µ�ת���ʴ���P2����P1С��P2��ѹǿΪP2ʱ����Y�㷴Ӧδ�ﵽƽ�⣬��Ӧ�����ƶ�������v��������v���棩����ͼ֪�����¶���X����������ƽ��ʱ�����ת����Ϊ50%��������ʽ��

�����ʵ���=0.3mol����ѹΪP2����ƽ�ⳣ��Kp=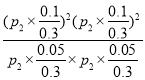 =
=![]() ��
��
�ʴ�Ϊ�����ڣ�![]() ��
��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�����Ŀ����һ���¶��£�10mL0.40mol/L H2O2�������ֽ⡣��ͬʱ�̲ⶨ����O2�������������Ϊ��״�������±���
t/min | 0 | 2 | 4 | 6 | 8 | 10 |
V(O2)/mL | 0.0 | 9.9 | 17.2 | 22.4 | 26.5 | 29.9 |
������������ȷ���ǣ���Һ����仯���Բ��ƣ�
A��0~6min��ƽ����Ӧ���ʣ�v��H2O2��![]()
![]() mol/(L��min)
mol/(L��min)
B��6~10min��ƽ����Ӧ���ʣ�v��H2O2����![]() mol/(L��min)
mol/(L��min)
C����Ӧ��6minʱ��c��H2O2��=0.3mol/L
D����Ӧ��6minʱ��H2O2�ֽ���50%