��Ŀ����
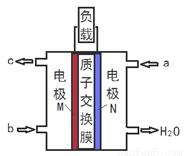
������������ͭ�Ƚ������仯�����ڹ�ҵ������Ҫ��;��
��1����Ԫ�ػ�̬ԭ�ӵĵ����Ų�ʽΪ_________������ͭ���ڰ�ˮ���γ�[Cu��NH3��4]SO4��Һ��[Cu��NH3��4]SO4�������ӵ����幹����______��_________���ܹ¶Ե��Ӷԣ�����ͭ�Ķѻ���ʽΪ______��
��2���Ʊ�Cr2O2Cl2�ķ�ӦʽΪK2Cr2O2+3CCl4=2KCl+2Cr2O2Cl2+3COCl2����
��������ѧ����ʽ�зǽ���Ԫ�ص縺����С�����˳����_____________����Ԫ�ط��ű�ʾ����
��COCl2����������ԭ�Ӿ�����8���ӹ��ͣ�COCl2����������ԭ�ӵ��ӻ���ʽΪ________��COCl2�����Ш����ͦм��ĸ�����Ϊ__________��
��NiO��FeO�ľ���ṹ�����Ȼ��Ƶľ���ṹ��ͬ������Ni2+��Fe2+�����Ӱ뾶�ֱ�Ϊ6.9��10-2nm��7.8��10-2nm�����۵㣺NiO_________FeO���>������<����=������
��4��Cu��Ca�ĺϽ�ʷ�ĩ״�����ױ��������侧���ṹ��ͼ��ʾ��
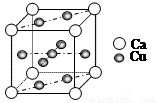
�ٸþ���Ļ�ѧʽΪ_________��ͭ�����ֽ������ͭ���۵�ȸƸߣ���ԭ����_______��
����֪�þ������ܶ�Ϊdg��cm-3��Ca�뾶Ϊr1cm��Cu�뾶Ϊr2cm����NAΪ����٤����������þ����Ŀռ�������Ϊ________���ú�d��r1��r2��NA�Ĵ���ʽ��ʾ����


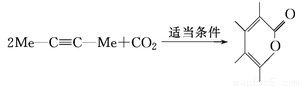

 ��Ϊͬ���칹��
��Ϊͬ���칹�� ����һ�ֻ�ѧ���Ժܸߵ�����������ú�����С�
����һ�ֻ�ѧ���Ժܸߵ�����������ú�����С� ��Ӧ����Ϊ ��
��Ӧ����Ϊ �� ����˫�����ϩ��ͬ���칹�壬����������ӽṹʾ��ͼ�мн�ֻ��135�㡢90�����֣�����һ�ȴ����� �֡�
����˫�����ϩ��ͬ���칹�壬����������ӽṹʾ��ͼ�мн�ֻ��135�㡢90�����֣�����һ�ȴ����� �֡�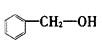 ���ڷ��࣬������Ϊ-OH
���ڷ��࣬������Ϊ-OH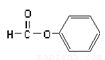 ����ȩ�࣬������Ϊ-CHO
����ȩ�࣬������Ϊ-CHO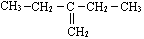 ������Ϊ��2-�һ�-1-��ϩ
������Ϊ��2-�һ�-1-��ϩ