��Ŀ����
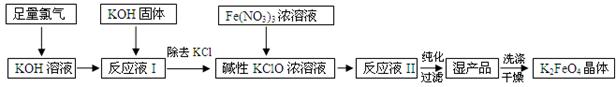
�������(K2FeO4)�����Ͷ��ˮ�����������������������ȶ����������������£�
��ش���������
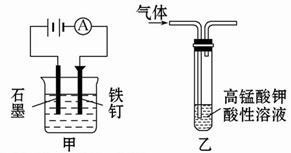
��1���ȼҵ��Cl2�Ļ�ѧ��Ӧ����ʽ ��
��2�����ɡ���ӦҺ�����ӷ���ʽ�� ����3��ʹ�ü���KClO��ԭ���� ��
��4���ӡ���ӦҺII���з����K2FeO4�� ������Ʒ��KCl�� (�ѧʽ)���û������� ���������ᴿ������ĸ��ţ���
��5����ҵ����3.96t K2FeO4������������Cl2�����ʵ���Ϊ______mol��

��ش���������
��1���ȼҵ��Cl2�Ļ�ѧ��Ӧ����ʽ ��
��2�����ɡ���ӦҺ�����ӷ���ʽ�� ����3��ʹ�ü���KClO��ԭ���� ��
��4���ӡ���ӦҺII���з����K2FeO4�� ������Ʒ��KCl�� (�ѧʽ)���û������� ���������ᴿ������ĸ��ţ���
| A������ | B����Һ | C������ | D���ؽᾧ |
��1��2NaCl + 2H2O 2NaOH + H2��+ Cl2��
2NaOH + H2��+ Cl2��
��2��3ClO- + 2Fe3+ + 10OH- = 2FeO42- + 3Cl- + 5H2O��
��3��K2FeO4�ڼ����������ȶ���KClO������ǿ��K2FeO4
��4�� KNO3 �� D
��5�� 3.00 �� 104
 2NaOH + H2��+ Cl2��
2NaOH + H2��+ Cl2�� ��2��3ClO- + 2Fe3+ + 10OH- = 2FeO42- + 3Cl- + 5H2O��
��3��K2FeO4�ڼ����������ȶ���KClO������ǿ��K2FeO4
��4�� KNO3 �� D
��5�� 3.00 �� 104
�����������1���ȼҵ���ǵ�ⱥ��ʳ��ˮ�õ��ռ����������2���������̷�����ӦҺII�����ô���������������ӵõ�FeO42-�����������ԭ��Ӧ���ӷ���ʽ����д����ɵã�3ClO- + 2Fe3+ + 10OH- = 2FeO42- + 3Cl- + 5H2O����3����Ŀ��Ϣ��K2FeO4�ڼ����������ȶ�����Ӧ���ڼ��������½��У���4����Ӧ�м����������������Ի�����KNO3������ص��ܽ�����¶ȱ仯�ϴ�������ȴ�ᾧ�ķ������룻��5������������Ӧ�ʹ����������������Ӧ�Ĺ�ϵʽ���м��㡣

��ϰ��ϵ�д�
�����Ŀ

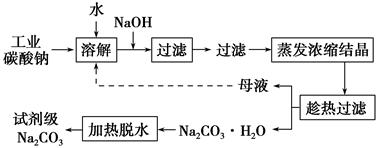
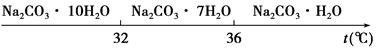
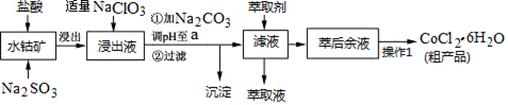
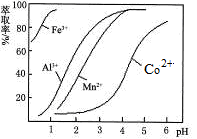
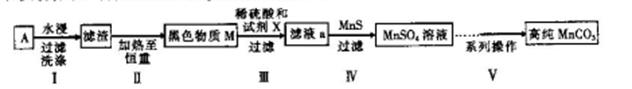
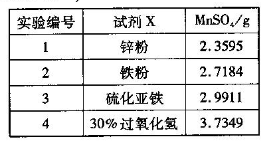
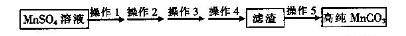
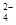 �ѳ��ɾ��� ����4��___________________��
�ѳ��ɾ��� ����4��___________________��