��Ŀ����
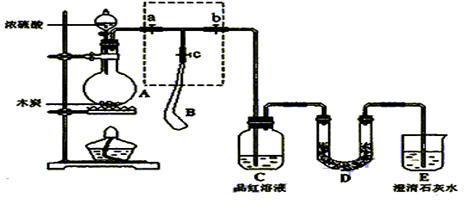
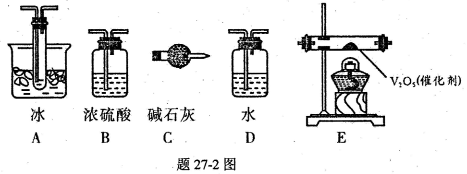
��14�֣�Ϊ����Ũ������ľ̿�ڼ��������·�Ӧ������SO2��CO2���壬�������ͼ��ʾʵ��װ�ã�a��b��cΪֹˮ�У�B�����ڴ��������ң�D�з��������Ը��������Һ��ʪ����֬�ޡ�
 ��ش��������⣺
��ش��������⣺
��1��װ��A��D�з�����Ӧ�Ļ�ѧ����ʽΪ �� ��
��2��ʵ��ǰ�����װ��A�������ԣ����Բ�ȡ�IJ����� ��
��3����ʵ��ɰܵĹؼ����ڿ��Ʒ�Ӧ������������ʲ��ܹ��죬����������� �ֵ�װ�ã�����ȷ�IJ���
�ֵ�װ�ã�����ȷ�IJ��� ˳���� ���ò��������д��
˳���� ���ò��������д��
����Aװ���м���Ũ���ᣬ���ȣ�ʹA�в����������������B���������г���һ��������ʱ��ֹͣ���ȣ�
�ڴ�װ��A��ȴ��������B��������ٱ仯�ر�ֹ ˮ��a����ֹˮ��b��������ѹ���ң�ʹ����B��������������װ��C�У����ﵽʵ��Ŀ�ĺر�ֹˮ��b��
ˮ��a����ֹˮ��b��������ѹ���ң�ʹ����B��������������װ��C�У����ﵽʵ��Ŀ�ĺر�ֹˮ��b��
�۴�ֹˮ��a��c���ر�ֹˮ��b��
��4��ʵ��ʱ��װ��C�е�����Ϊ ��
��5����D�в��� ����ʱ������˵��ʹE�г���ʯ��ˮ����ǵ���CO2��������SO2��
��6��װ��D������Ϊ ��
|
 ��ش��������⣺
��ش��������⣺ ��1��װ��A��D�з�����Ӧ�Ļ�ѧ����ʽΪ �� ��
��2��ʵ��ǰ�����װ��A�������ԣ����Բ�ȡ�IJ����� ��
��3����ʵ��ɰܵĹؼ����ڿ��Ʒ�Ӧ������������ʲ��ܹ��죬�����������
 �ֵ�װ�ã�����ȷ�IJ���
�ֵ�װ�ã�����ȷ�IJ��� ˳���� ���ò��������д��
˳���� ���ò��������д������Aװ���м���Ũ���ᣬ���ȣ�ʹA�в����������������B���������г���һ��������ʱ��ֹͣ���ȣ�
�ڴ�װ��A��ȴ��������B��������ٱ仯�ر�ֹ
 ˮ��a����ֹˮ��b��������ѹ���ң�ʹ����B��������������װ��C�У����ﵽʵ��Ŀ�ĺر�ֹˮ��b��
ˮ��a����ֹˮ��b��������ѹ���ң�ʹ����B��������������װ��C�У����ﵽʵ��Ŀ�ĺر�ֹˮ��b���۴�ֹˮ��a��c���ر�ֹˮ��b��
��4��ʵ��ʱ��װ��C�е�����Ϊ ��
��5����D�в��� ����ʱ������˵��ʹE�г���ʯ��ˮ����ǵ���CO2��������SO2��

��6��װ��D������Ϊ ��
��1��C +2H2SO4��Ũ��
+2H2SO4��Ũ��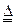 CO2��+2SO2��+2H2O 2KMnO4+5SO2+2H2O=K2SO4+2MnSO4+2H2HO4����2�֣�
CO2��+2SO2��+2H2O 2KMnO4+5SO2+2H2O=K2SO4+2MnSO4+2H2HO4����2�֣�
��2���رշ�Һ©���Ļ�������ֹˮ��a��b���ر�ֹˮ��c���������ȣ����ȣ�Բ����ƿA����C��������ð�����ƿ����ƣ�����Դ����C����������ˮ��������֤��Aװ�õ����������á���2�֣�
��3���ۢ٢� ��2�֣�
��4�� Ʒ����Һ��ɫ����2�֣�
��5��������һ����֬����ɫ��dz��������һ����֬����ɫ���䣻��2�֣�
��6����ȥSO2������SO2�ѱ���������2�֣�
 +2H2SO4��Ũ��
+2H2SO4��Ũ��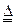 CO2��+2SO2��+2H2O 2KMnO4+5SO2+2H2O=K2SO4+2MnSO4+2H2HO4����2�֣�
CO2��+2SO2��+2H2O 2KMnO4+5SO2+2H2O=K2SO4+2MnSO4+2H2HO4����2�֣���2���رշ�Һ©���Ļ�������ֹˮ��a��b���ر�ֹˮ��c���������ȣ����ȣ�Բ����ƿA����C��������ð�����ƿ����ƣ�����Դ����C����������ˮ��������֤��Aװ�õ����������á���2�֣�
��3���ۢ٢� ��2�֣�
��4�� Ʒ����Һ��ɫ����2�֣�
��5��������һ����֬����ɫ��dz��������һ����֬����ɫ���䣻��2�֣�
��6����ȥSO2������SO2�ѱ���������2�֣�
��

��ϰ��ϵ�д�
�����Ŀ
 ����KMnO4��Һb mL��
����KMnO4��Һb mL��
 ���
��� ��
��
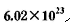 ������ת�ƣ���÷�Ӧ�Ļ�ѧ����ʽΪ__________________
������ת�ƣ���÷�Ӧ�Ļ�ѧ����ʽΪ__________________ ��ĩ������Ũ������д�ʵ�飬����Ӧ����ʱ������ͨ��O2 ����ʱ����װ��IV������ mg,��ʵ����SO2��ת����Ϊ__________________.(�ú�m��n�Ĵ���ʽ��д��
��ĩ������Ũ������д�ʵ�飬����Ӧ����ʱ������ͨ��O2 ����ʱ����װ��IV������ mg,��ʵ����SO2��ת����Ϊ__________________.(�ú�m��n�Ĵ���ʽ��д��