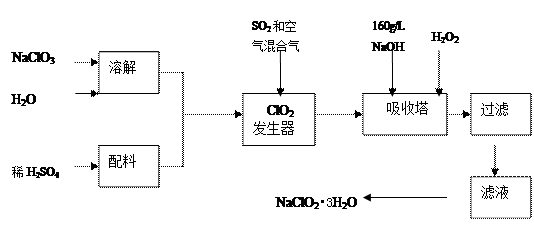
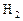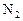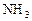��Ŀ����
��7�֣��״��ǿ�����ȼ�ϣ�����������Ȼ�������������������ɱ��ͣ���;�㷺������п�ѧ��������Ѻ���0��050%CO2�Ŀ�����ͨ������¿�����CO2���������Ϊ0��030%����������CO2�������������0��050%ʱ�����������Ե�����ЧӦ��������̼�����Һ�У�Ȼ�����뷨��CO2����Һ����ȡ����������ѧ��Ӧ��õ��״����乹�뼼���������£�
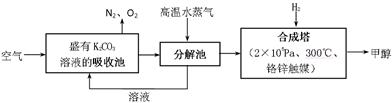
�Իش��������⣺
��1��д�����ճ�����Ҫ��Ӧ�����ӷ���ʽ��
____________________ __ _��
��2��д���ϳ����з�Ӧ�Ļ�ѧ����ʽ��
_________________ _ __��
��3��������Ŀ������Ϊ33��6m3����״���£������������������� g�״�

�Իش��������⣺
��1��д�����ճ�����Ҫ��Ӧ�����ӷ���ʽ��
____________________ __ _��
��2��д���ϳ����з�Ӧ�Ļ�ѧ����ʽ��
_________________ _ __��
��3��������Ŀ������Ϊ33��6m3����״���£������������������� g�״�
��7�֣���1��CO32- + CO2 + H2O="=" 2HCO3- ��2�֣�
��2��CO2 + 3H2 ="=" CH3OH + H2O ��2�֣�
��3��24 ��3�֣�
��2��CO2 + 3H2 ="=" CH3OH + H2O ��2�֣�
��3��24 ��3�֣�
��

��ϰ��ϵ�д�
 ������ӱ������ͯ������ϵ�д�
������ӱ������ͯ������ϵ�д� A�ӽ��� ϵ�д�
A�ӽ��� ϵ�д� ȫ�Ų��Ծ�ϵ�д�
ȫ�Ų��Ծ�ϵ�д�
�����Ŀ


 2NH3��g������
2NH3��g������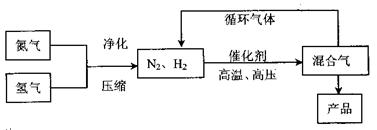
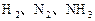 ��Ӧ��ƽ��ʱ������Ũ������һ���ǣ� ��
��Ӧ��ƽ��ʱ������Ũ������һ���ǣ� ��