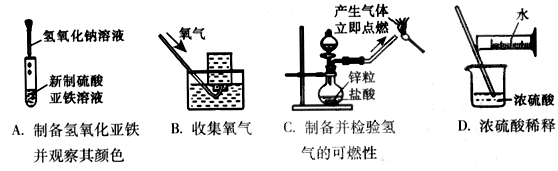
��Ŀ����
�Ȼ������ε�ⷨ�����Ȼ���Ϊԭ�ϣ��Լ��������������Ȼ��������MgCl2��KCl��CaCl2��Ϊ����ʽ��е����ȡ���ķ�����
��1���Ȼ������ε�ⷨ��Ҫ���ƴ�������������������Ҫ�ɷ�ΪAl2O3����������Fe2O3��SiO2�����ʣ�Ϊԭ��ͨ������;���ᴿ��������

������д��X��Y�ijɷ� �� ��
����ͼ��ʾ��ʵ�����н��й��ˣ������е���������ֱ��� �� ��
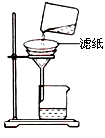
��2���Ʊ���ˮ�Ȼ����ķ�ӦΪ��2Al2O3+6Cl2 4AlCl3+3O2
4AlCl3+3O2
��Ϊ�ٽ��÷�Ӧ�Ľ��У�ʵ������������뽹̿����ԭ���� ��
�ܼ��뽹̿��Ļ�ѧ��Ӧ�ɱ�ʾΪAl2O3+C+Cl2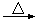 AlCl3+X����Ϊȷ������X�Ƿ��ǻ�����壬ijͬѧ��X����ͨ�����ȵ�����ͭ�ͳ����ʯ��ˮ���ٸ��������жϡ��ò����Ƿ���ȷ��������ȷ������ȷ�����жϣ� ����˵������ ��
AlCl3+X����Ϊȷ������X�Ƿ��ǻ�����壬ijͬѧ��X����ͨ�����ȵ�����ͭ�ͳ����ʯ��ˮ���ٸ��������жϡ��ò����Ƿ���ȷ��������ȷ������ȷ�����жϣ� ����˵������ ��
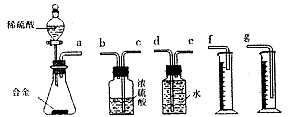
��3�����ڹ�ҵ��ͨ���õ��������������ʽ��ȡ��������������1�������ĵ�������������_________
������ڡ�����С�ڡ��������ڡ����Ȼ���������
��1���Ȼ������ε�ⷨ��Ҫ���ƴ�������������������Ҫ�ɷ�ΪAl2O3����������Fe2O3��SiO2�����ʣ�Ϊԭ��ͨ������;���ᴿ��������

������д��X��Y�ijɷ� �� ��
����ͼ��ʾ��ʵ�����н��й��ˣ������е���������ֱ��� �� ��

��2���Ʊ���ˮ�Ȼ����ķ�ӦΪ��2Al2O3+6Cl2
 4AlCl3+3O2
4AlCl3+3O2��Ϊ�ٽ��÷�Ӧ�Ľ��У�ʵ������������뽹̿����ԭ���� ��
�ܼ��뽹̿��Ļ�ѧ��Ӧ�ɱ�ʾΪAl2O3+C+Cl2
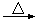 AlCl3+X����Ϊȷ������X�Ƿ��ǻ�����壬ijͬѧ��X����ͨ�����ȵ�����ͭ�ͳ����ʯ��ˮ���ٸ��������жϡ��ò����Ƿ���ȷ��������ȷ������ȷ�����жϣ� ����˵������ ��
AlCl3+X����Ϊȷ������X�Ƿ��ǻ�����壬ijͬѧ��X����ͨ�����ȵ�����ͭ�ͳ����ʯ��ˮ���ٸ��������жϡ��ò����Ƿ���ȷ��������ȷ������ȷ�����жϣ� ����˵������ ����3�����ڹ�ҵ��ͨ���õ��������������ʽ��ȡ��������������1�������ĵ�������������_________
������ڡ�����С�ڡ��������ڡ����Ȼ���������
��10�֣���1����NaOH��Һ��1�֣��� ϡ���ᣨ1�֣�
��û��ʹ�ò�������������ֽ��Ե����©����Ե��2�֣�
��2����̼��O2��Ӧ�������ڷ�Ӧ������У�2�֣�
�ܲ���ȷ��������������ͨ�����ȵ�CuO��ĩ������CO�ͻ�����CO2����������ȷ��ԭ���Ƿ���CO2����2�֣� ��3��С�ڣ�2�֣�
��û��ʹ�ò�������������ֽ��Ե����©����Ե��2�֣�
��2����̼��O2��Ӧ�������ڷ�Ӧ������У�2�֣�
�ܲ���ȷ��������������ͨ�����ȵ�CuO��ĩ������CO�ͻ�����CO2����������ȷ��ԭ���Ƿ���CO2����2�֣� ��3��С�ڣ�2�֣�
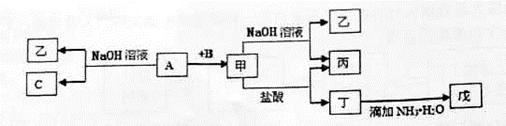
�����������1������Һ���ܺͰ�ˮ��Ӧ��������������������˵����Һ���к��������ӣ�����YӦ�������ᡣ����Һ���к���AlO2����SiO32�������X��NaOH�����������������Ʋ���Ӧ�������õ��������������ƺ����ᷴӦ���ɹ�������������õ��Ȼ���������Ļ��Һ�����백����������������������
�ڸ���װ��ͼ��֪����װ���ǹ���װ�ã������Ҫ�Ĵ�������û��ʹ�ò����������������ֽ��Ե����©����Ե��
��2������Ϊ�ڼ��ȵ������£�̼��O2��Ӧ������CO��CO2������������Ũ�ȣ������ڷ�Ӧ������С�
�������ڼ��ȵ������£�CO�ܺ�����ͭ��Ӧ����CO2�����Խ�����������ͨ�����ȵ�CuO��ĩ������CO�ͻ�����CO2����������ȷ��ԭ���Ƿ���CO2�������Dz���ȷ�ġ�
��3������ԭ���غ��֪��1mol��������������2mol�Ȼ�����1mol��������������102g��2mol�Ȼ�����������2mol��133.5g/mol��267g������������������С���Ȼ�����������

��ϰ��ϵ�д�
 ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д�
ӥ�ɽ̸��νӽ̲ĺӱ�����������ϵ�д� ���������ν�ϵ�д�
���������ν�ϵ�д�
�����Ŀ
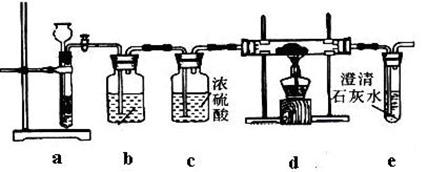
 ������ȷ���ܴﵽĿ�ĵ���
������ȷ���ܴﵽĿ�ĵ���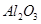 ����
����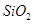 ��
��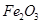 ��MgO�����ʣ�����ȡ���ֹ���Ʒ���������£�
��MgO�����ʣ�����ȡ���ֹ���Ʒ���������£�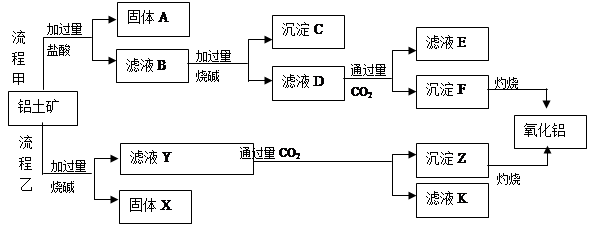
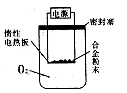
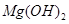 ���ܶȻ�����
���ܶȻ�����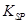 =10-11,ȡ��������ҺB,����һ�������ռ�ǡʹþ���ӳ�����ȫ������Һ��PH��СΪ_______.
=10-11,ȡ��������ҺB,����һ�������ռ�ǡʹþ���ӳ�����ȫ������Һ��PH��СΪ_______.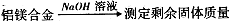 ��
��
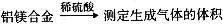 ��
��
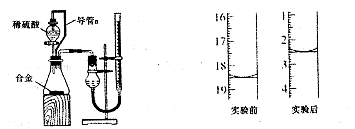
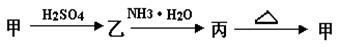
 ��ת�������ӷ���ʽ ��________________________ ��
��ת�������ӷ���ʽ ��________________________ ��