��Ŀ����
�����ε�ⷨ��õĴ�������һ�����Ľ����ƺ���������Щ���ʿɲ��ô�����������ȥ��������β��������������ڸղŶ����������������£�
��ע ��NaCl�۵�Ϊ801�棻AlCl3��181��������
��NaCl�۵�Ϊ801�棻AlCl3��181��������
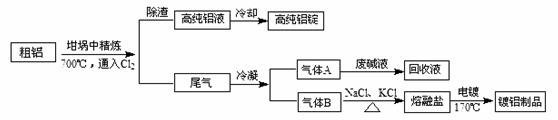
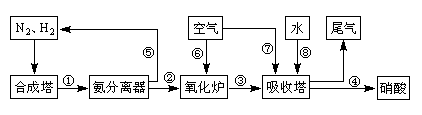
��1������ǰ������������������������ʯӢɰ����ֹ����ʱ���Ƿֱ����������û���Ӧ�����µ����ʣ���صĻ�ѧ����ʽΪ�� �͢�
��2����Cl2����ͨ�������еĴ������壬�����������ϸ���ȥ�����ݵ���Ҫ�ɷֳ�Cl2����� ����̬����ճ���������ϣ�����������γɸ����������п϶�����
��3�����÷ϼ�Һ����A�Ĺ����У���������Ӧ�����ӷ���ʽΪ
��4�����������У�������Ϊ ���������ε������Ԫ�غ���Ԫ����Ҫ��AlCl4�D ��Al2Cl7�D��ʽ���ڣ�����Ƶ���Ҫ�缫��ӦʽΪ
��5���ֲĶ��������γɵ�����������Ĥ�ܷ�ֹ�ֲĸ�ʴ����ԭ����

��ע
 ��NaCl�۵�Ϊ801�棻AlCl3��181��������
��NaCl�۵�Ϊ801�棻AlCl3��181����������1������ǰ������������������������ʯӢɰ����ֹ����ʱ���Ƿֱ����������û���Ӧ�����µ����ʣ���صĻ�ѧ����ʽΪ�� �͢�
��2����Cl2����ͨ�������еĴ������壬�����������ϸ���ȥ�����ݵ���Ҫ�ɷֳ�Cl2����� ����̬����ճ���������ϣ�����������γɸ����������п϶�����
��3�����÷ϼ�Һ����A�Ĺ����У���������Ӧ�����ӷ���ʽΪ
��4�����������У�������Ϊ ���������ε������Ԫ�غ���Ԫ����Ҫ��AlCl4�D ��Al2Cl7�D��ʽ���ڣ�����Ƶ���Ҫ�缫��ӦʽΪ
��5���ֲĶ��������γɵ�����������Ĥ�ܷ�ֹ�ֲĸ�ʴ����ԭ����
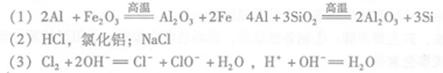
(4) �� Al-3e-+7AlCl4-="===" 4Al2Cl7-

��

��ϰ��ϵ�д�
�����Ŀ
 CO2+H2��t��ʱ����1L�ܱ������г���0.2mol CO��0.3molˮ��������Ӧ����ƽ�����ϵ��c(H2)=0.12mol��L-1�����¶��´˷�Ӧ��ƽ�ⳣ��K=_____�������������
CO2+H2��t��ʱ����1L�ܱ������г���0.2mol CO��0.3molˮ��������Ӧ����ƽ�����ϵ��c(H2)=0.12mol��L-1�����¶��´˷�Ӧ��ƽ�ⳣ��K=_____�������������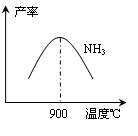
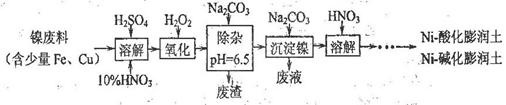
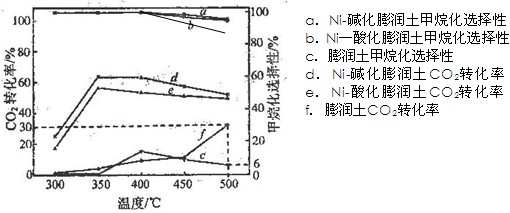
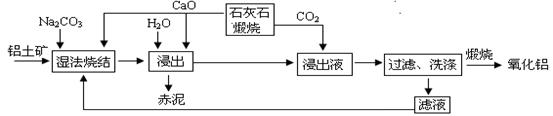
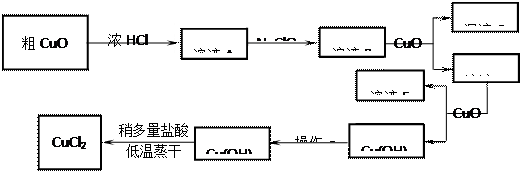
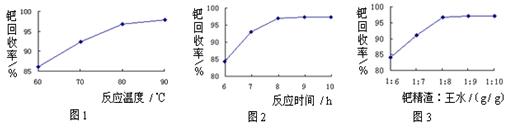
 ����̿������Ԫ��������þ�������衢ͭ�ȣ�����ȡ�����٣���Pd��99.9%���IJ��ֹ����������£�
����̿������Ԫ��������þ�������衢ͭ�ȣ�����ȡ�����٣���Pd��99.9%���IJ��ֹ����������£�